3DHEALS March 2026 Roundup Covers FDA Clearances, Bioprinted Tissue, and AI Quality Control
FDA clearances for AM devices are quietly rewriting the materials rulebook — and 3DHEALS's March 2026 Lattice episode explains why your next filament choice may benefit.

The moment an FDA 510(k) clearance lands for an additively manufactured device, the regulatory playbook for every comparable AM workflow shifts with it. That is the central thread running through 3DHEALS's March 29 Lattice episode, which surveyed the month's most consequential developments across clinical additive manufacturing: new FDA clearances resetting baseline expectations for implantable devices, bioprinted tissue models applied to live oncology research, point-of-care printing wins inside hospital systems, and a frank reassessment of what AI can and cannot yet deliver in regulated clinical environments.
Allumin8's FDA 510(k) clearance for the A8 INTEGR8 Porous Pedicle Screw System, earned after a six-year development cycle built around bone ingrowth geometry and fatigue performance, illustrates the kind of precedent the episode is tracking. Alyssa Huffman's St. Louis company produced the world's first 5.5mm pedicle screw via additive manufacturing to clear the agency's review, and the documented validation trail that clearance generates reaches beyond that single product. When the FDA approves a specific AM process for a load-bearing implantable device, it creates a reference framework that research institutions, point-of-care facilities, and informed makers working in prosthetics or surgical training aids can draw from directly.
The AI coverage in the episode was notably measured. While AI-driven defect detection and automated process qualification are accelerating how hospitals validate in-house printing workflows, the episode flagged the persistent tension between technological enthusiasm and clinical reality: AI can automate layer-by-layer inspection and flag parameter drift in real time, but the hosts stressed that human oversight and documented validation remain non-negotiable when patient safety is involved. Hype is moving faster than clearance timelines, and the episode treated that gap as a warning, not a footnote.
That clinical caution does not translate into a slow rollout on the consumer side. Platforms like SimplyPrint have trained computer vision systems on more than five million hours of print data since 2020, while OctoEverywhere's Gadget tool uses webcam feeds to catch adhesion failures and spaghetti mid-print. Obico's failure detection runs on a $30 Raspberry Pi paired with a basic webcam. These tools are not waiting for industrial-grade AI to trickle down; they represent a parallel development track that the maker community is building for itself, and they are maturing quickly.
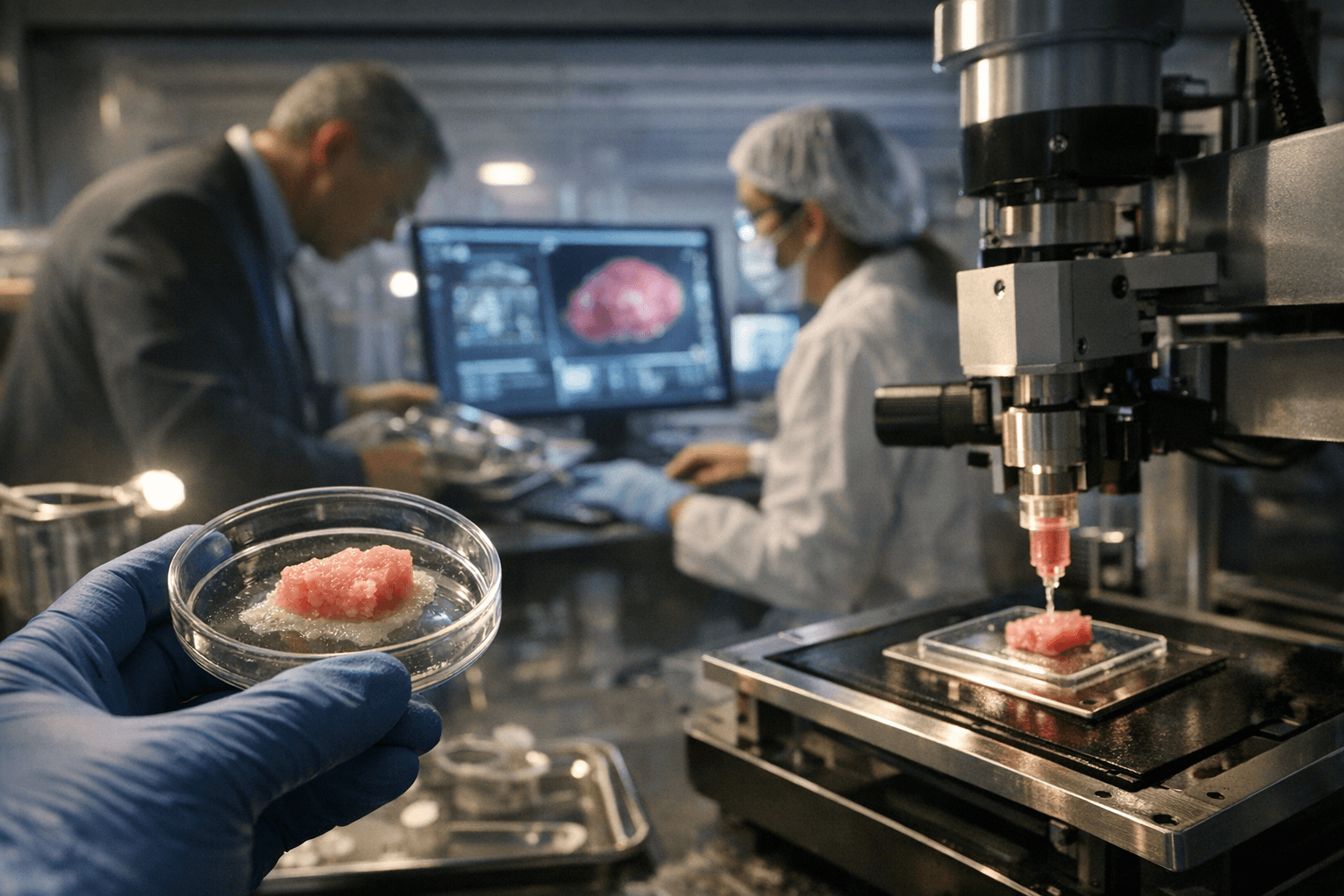
The point-of-care dimension of the March episode carries the most direct relevance for anyone printing anatomical models, orthotics, or surgical training aids. Hospital programs that built validated in-house workflows reported measurably shorter clinical timelines, an outcome only achievable when materials and processes carry documented performance data. For makers selecting filaments for prosthetic liners or resins for anatomical models, FDA-cleared AM workflows translate into one concrete and often overlooked benefit: they specify which biocompatibility standard a material was actually tested against. A datasheet that claims "biocompatible" without citing ISO 10993 or a comparable framework offers no real safety assurance, while a cleared device's documentation does.
The March Lattice episode's value is not in the individual headlines it covers but in what those headlines collectively confirm: healthcare AM is graduating from isolated research demonstrations to regulated, reproducible clinical workflows, and the materials and process standards being written at that level will define responsible practice at every level below it.
Know something we missed? Have a correction or additional information?
Submit a Tip

