Bavarian Nordic raises 2026 guidance after $97 million U.S. vaccine option
Bavarian Nordic lifted 2026 guidance after the U.S. added a $97 million smallpox vaccine option, signaling stockpile spending still drives biodefense procurement.
Bavarian Nordic lifted its 2026 outlook after the U.S. government added a $97 million option to its smallpox vaccine contract, a move that points less to an immediate outbreak response than to a continuing federal push to keep biodefense stockpiles ready.
The Danish vaccine maker said the extra option, from the U.S. Department of Health and Human Services, was tied to the freeze-dried form of JYNNEOS, its mpox and smallpox vaccine. The company raised its full-year revenue forecast to 5.5 billion to 5.7 billion Danish crowns, up from 5 billion to 5.2 billion crowns, and increased its EBITDA margin forecast to about 28% from 25%.

The latest U.S. commitment builds on a broader contract structure managed by the Biomedical Advanced Research and Development Authority, which sits within HHS’s Administration for Strategic Preparedness and Response. Bavarian Nordic said BARDA had already exercised additional options valued at $143.6 million under the existing contract in May 2025 to supply freeze-dried JYNNEOS to the United States in 2026, with deliveries planned for that year. The company said the freeze-dried formulation received FDA approval in March 2025.
That formulation matters because it is designed for stockpiling. Bavarian Nordic said freeze-dried JYNNEOS improves transportation, storage conditions and shelf life, all critical advantages for federal reserves that may sit unused for long periods before being deployed. ASPR says the Strategic National Stockpile holds large quantities of medicines, vaccines and other supplies for chemical, biological, radiological and nuclear threats, as well as emerging infectious diseases and pandemics.
Bavarian Nordic has worked with the U.S. government on smallpox vaccine development, manufacturing and supply since 2003. The company says JYNNEOS was approved in 2019 and was the first smallpox vaccine successfully developed under Project BioShield, the federal program created to speed countermeasure development for major public-health threats.
The procurement pattern also shows that U.S. demand has remained durable even as the worst of the mpox surge faded. Bavarian Nordic said in preliminary February 2026 results that 2025 revenue reached 6,244 million Danish crowns, including 3,105 million from Public Preparedness, a figure it said exceeded its normal annual base business by more than 1,000 million crowns because mpox vaccine demand continued last year. The company also said in February that it had secured a $22.5 million contract from the Government of Canada for its mpox and smallpox vaccine.
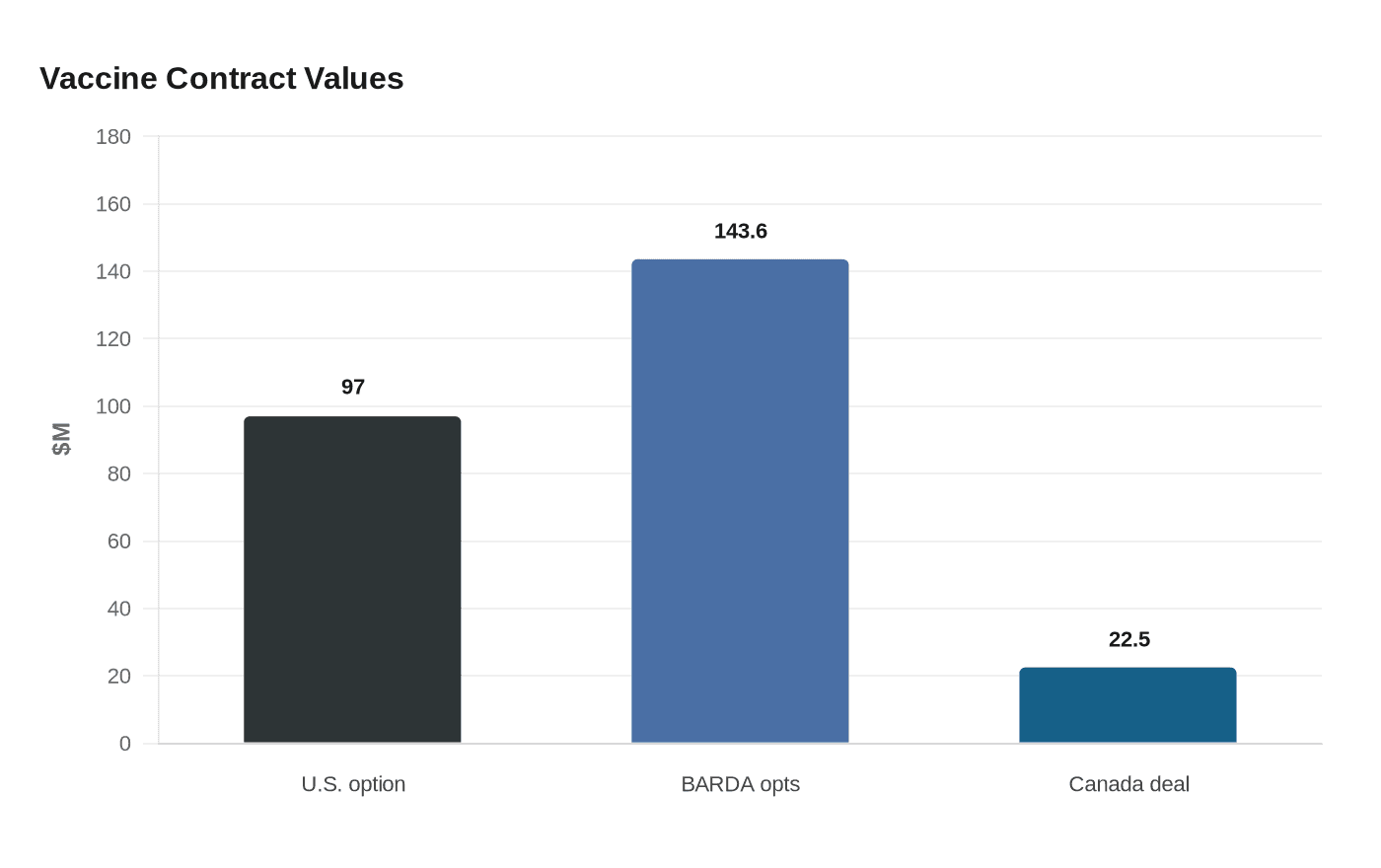
For Washington, the message is clear: federal vaccine spending is still being shaped by long-horizon preparedness planning, with the Strategic National Stockpile and BARDA continuing to favor supplies that can be held in reserve and deployed when the threat environment changes.
Know something we missed? Have a correction or additional information?
Submit a Tip

