Bristol Myers Squibb Partners With Oxford BioTherapeutics to Accelerate Oncology Drug Discovery
Oxford BioTherapeutics' OGAP-Verify platform, already behind three clinical programs, will supply BMS with next-gen T-cell engager candidates targeting solid tumors.

Bristol Myers Squibb has entered a multi-year selective target collaboration with Oxford BioTherapeutics to develop next-generation T-cell engager therapies for solid tumors, the Oxford, U.K.-based biotech disclosed on April 9. The deal gives BMS access to a target-discovery platform that has already delivered three programs to clinical stage and grants Oxford BioTherapeutics a path through clinical development without bearing the full cost of that journey alone.
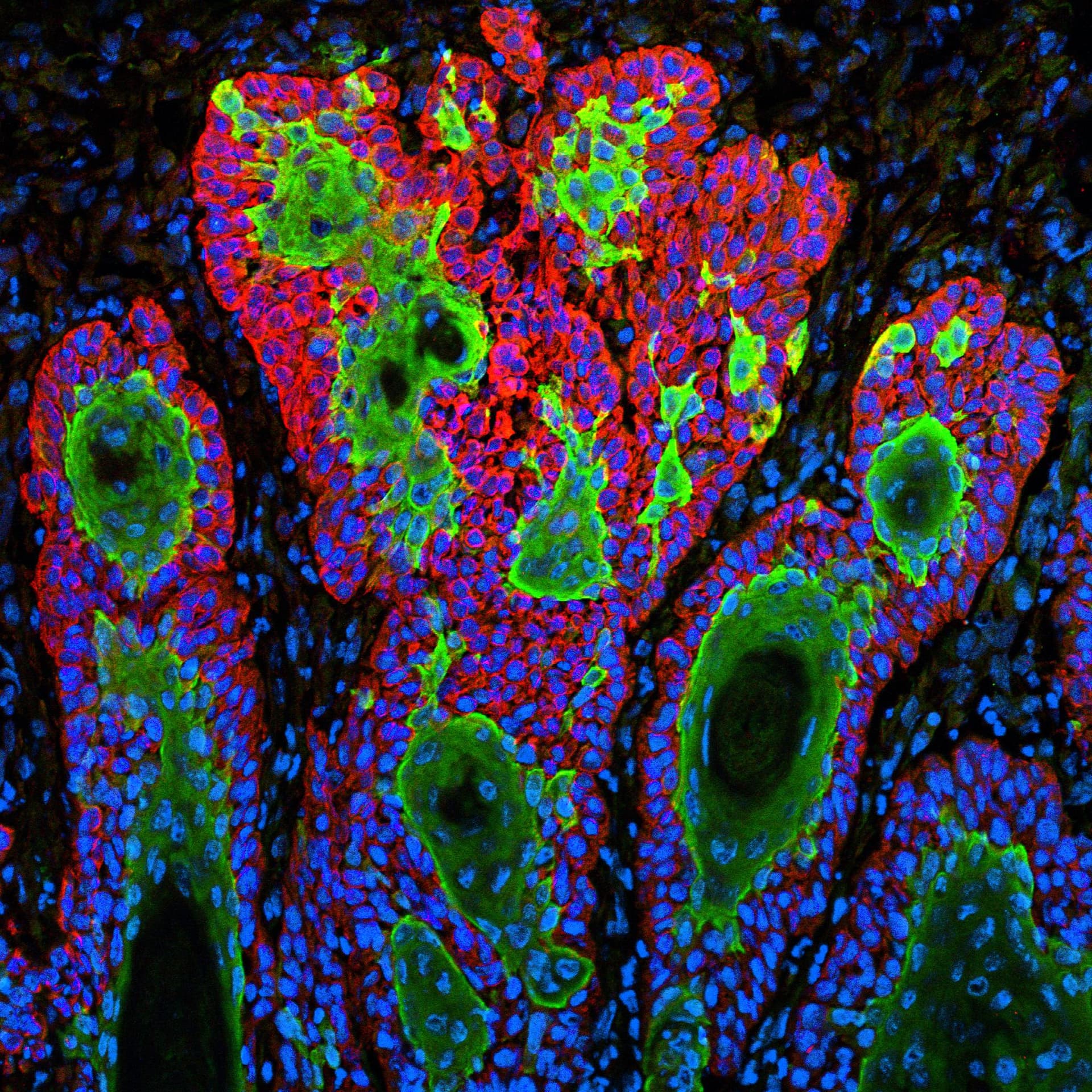
At the center of the arrangement is Oxford BioTherapeutics' OGAP-Verify platform, a proprietary system designed to identify tumor-selective antigens with heightened sensitivity and specificity compared with conventional approaches. Identifying the right tumor antigen is one of the most consequential and failure-prone steps in oncology drug development: a target that appears compelling in laboratory assays but is also expressed on healthy tissue can doom a therapy to off-tumor toxicity before it ever reaches late-stage trials. OGAP-Verify attempts to close that gap by producing more precise target profiles before a molecule enters the costly preclinical process.
Under the terms announced, Oxford BioTherapeutics will go further than pure target nomination. The company will design and deliver development candidates, an expanded scope that reflects its evolution into what both parties characterized as a fully integrated discovery and preclinical development platform. BMS then assumes responsibility for clinical development and commercialization of any resulting therapies. Financial terms, including upfront payments and milestone values, were not disclosed.
"Collaborating with Bristol Myers Squibb, a global leader in oncology, represents an important milestone for OBT," said Christian Rohlff, PhD, chief executive of Oxford BioTherapeutics. The deal is the Oxford company's third partnership with a major pharmaceutical company in the past 12 months, following strategic agreements with GSK and Roche in 2025. Prior collaborators also include Boehringer Ingelheim and Zymeworks.
The therapeutic modality in focus, T-cell engagers, has drawn heavy investment across the industry for its ability to redirect a patient's own immune cells against tumors. Bi- and tri-specific formats hold particular promise for solid tumors, which have resisted many conventional immunotherapy approaches. The modality also carries a daunting safety constraint: molecules potent enough to kill tumor cells must avoid triggering T cells against normal tissue, a balance that has caused clinical failures for multiple programs.
For BMS, whose portfolio spans checkpoint inhibitors and cell therapies, the collaboration fits a pattern of externalized R&D, acquiring pre-validated targets and preclinical candidates from specialized platforms rather than originating every program internally. That model spreads early discovery risk, preserves resources for later-stage work, and can shorten the timeline to first-in-human studies. The BMS deal arrived as part of a broader wave of platform-plus-big-pharma arrangements announced in recent months as the industry accelerates investment in next-generation immunotherapy.
Three programs sourced from OGAP-Verify technology are already in clinical development in the United States and Europe, a track record Oxford BioTherapeutics is deploying to attract larger partners. Whether the BMS collaboration adds to that count depends on whether candidates clear preclinical and early clinical hurdles, particularly the on-tumor versus off-tumor selectivity challenge that defines the risk profile of every T-cell engager program that enters the clinic.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip