EU health chief urges collective drug buying to secure access to new medicines
Europe’s patients are waiting 578 days on average for approved medicines as Brussels weighs a bigger role in collective drug buying.

Emer Cooke pressed the European Union to go further in buying medicines together, arguing that collective procurement could help secure access to newly approved drugs as launches in Europe slow and patients wait longer for treatment.
The European Medicines Agency executive director said the bloc needs a broader mandate to act at the regional level, rather than leaving smaller countries to negotiate alone in a market shaped by pricing pressure and commercial strategy. Speaking at the Reuters Pharma Europe 2026 event in Barcelona, Cooke said governments were feeling the strain of U.S. policy changes and the resulting shifts in launch decisions. “Everybody is struggling with the consequences of U.S. pricing policy,” she said.

The pressure is visible in access data. EFPIA said the average time from approval to patient access across Europe was 531 days in 2024, then rose to 578 days in its 2025 update. In the EU average across 27 member states, only 46% of centrally approved innovative medicines were available to patients, down from 48% in 2019. Only 29% were fully available through public reimbursement, down from 42% in 2019, while 17% were available only with restrictions, up from 6%. EFPIA also showed how uneven access remains inside the bloc, with Germany at 126 days from approval to access and Poland at 804 days.
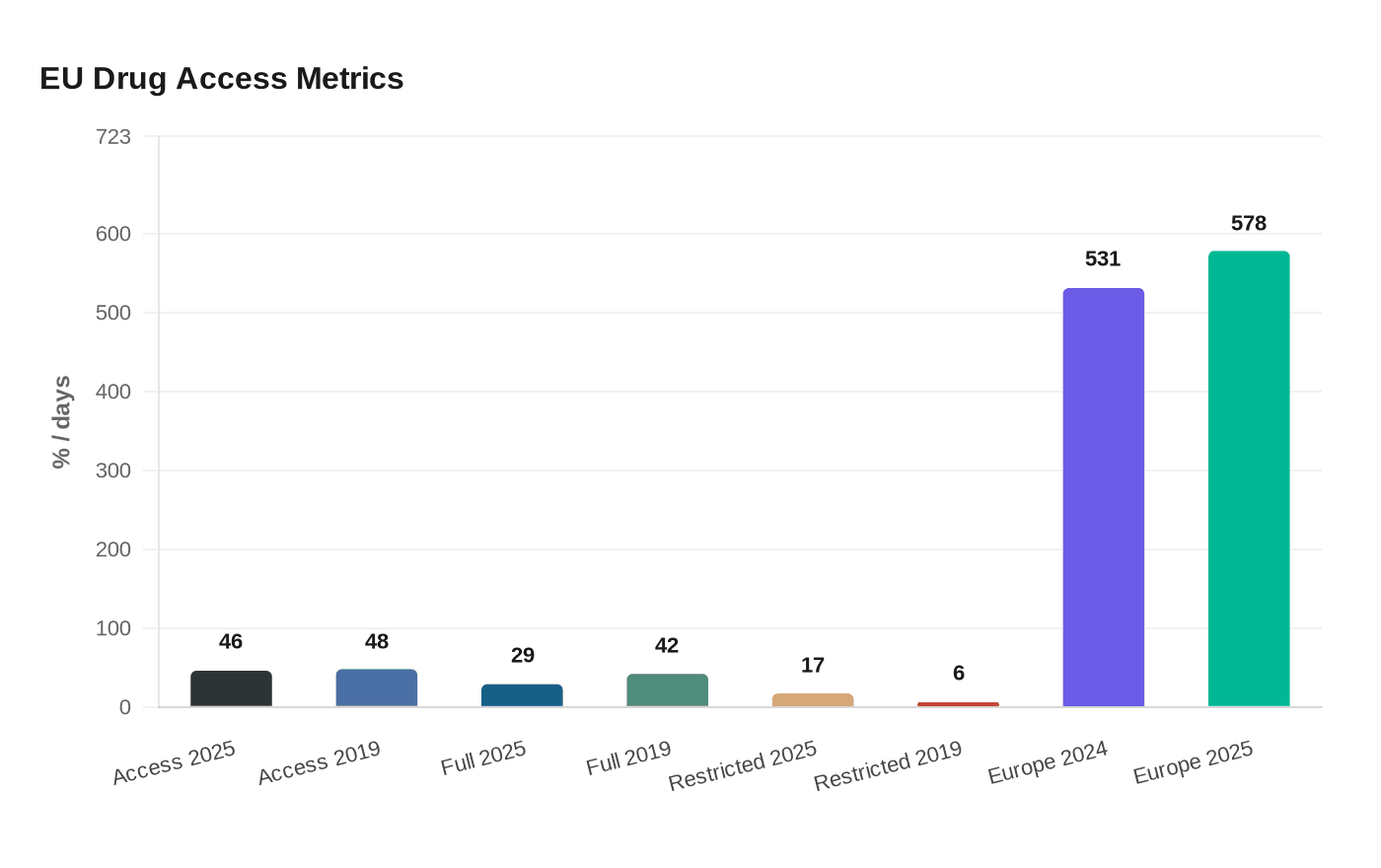
Those numbers have sharpened a debate already underway in Brussels over whether Europe should rely on fragmented national purchasing decisions or use more unified procurement tools to protect access. The European Commission proposed a Critical Medicines Act on March 11, 2025, saying it would support collaborative procurement among member states at their request to address availability gaps for critical medicines and other medicines of common interest. The Commission says the plan is also meant to diversify supply chains, strengthen EU pharmaceutical manufacturing and reduce dependence on single suppliers and third countries.
The wider reform of EU pharmaceutical legislation adds to that shift. The European Medicines Agency has called it the most significant overhaul of the regulatory framework in more than two decades, while the Council of the European Union said on June 4, 2025, that its position on the pharma package was aimed at ensuring timely and equitable access to safe, affordable and effective medicines. Europe has already used joint procurement in health emergencies, including a recent diphtheria therapeutic framework involving eight member states and 1,600 vials. Moldova joined the EU Joint Procurement Agreement in July 2024.
Cooke’s intervention puts a central policy choice back on the table: whether Europe can preserve national control while still pooling enough bargaining power to keep new medicines reaching patients across the bloc.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

