FDA clears JUXTAPID for children 2 and older with HoFH
Chiesi won FDA approval March 3 to expand JUXTAPID to children aged 2+, based on trial data and modeling, offers large LDL cuts but carries a liver toxicity risk.

Chiesi Global Rare Diseases said the Food and Drug Administration has approved a label expansion for JUXTAPID (lomitapide) capsules to treat pediatric patients aged 2 years and older with homozygous familial hypercholesterolemia, the company announced March 3. The move gives U.S. clinicians the first explicit approval to start lomitapide in very young children with the ultra-rare, genetically driven condition that produces dangerously high LDL cholesterol.
The pediatric approval rests on a 104-week single-arm trial, APH-19 (NCT04681170), that enrolled 43 children ages 5 to 17 and completed 39 participants. At the 24-week primary endpoint the trial reported a mean reduction in LDL-C of 49 percent from baseline, with a 95 percent confidence interval of minus 59 to minus 38 percent. Doses in the study began with age-dependent starting levels and were escalated to each patient’s maximum tolerated dose to meet LDL goals.
Chiesi said an integrated model-based analysis extended dose justification to children older than 2 years, allowing the FDA to clear the label for ages 2 and up despite the trial’s direct clinical data covering ages 5 to 17. Mitch Goldman, Senior Vice President of R&D at Chiesi Global Rare Diseases, framed the decision as a step for families: “This approval represents more than a regulatory milestone; it’s a meaningful advancement for children and families living with HoFH.” He added, “By expanding access to JUXTAPID for children 2 years of age and older, we’re enabling very young members of the HoFH community to benefit from the same proven treatment that has already helped adults manage their condition.”
The approval follows an earlier international milestone: Saudi Arabia’s SFDA in December granted pediatric authorization for the same drug, under the local name Lojuxta, for children aged 5 and older. BioSpace reported the Saudi assessment concluded reductions in LDL-C exceeded 50 percent at 24 weeks.
Regulators and physicians will weigh the drug’s substantial LDL lowering against its safety profile. U.S. prescribing information carries a boxed warning for hepatotoxicity. In the pediatric trial 6 of 43 patients, or about 14 percent, experienced elevations in alanine aminotransferase and/or aspartate aminotransferase at or above three times the upper limit of normal. Company materials report a median absolute increase in hepatic fat of 4 percentage points in pediatric patients over 24 and 104 weeks, from a 3 percent baseline. In adults the median hepatic fat increase was reported as 6 percentage points from 1 percent baseline at several time points measured by magnetic resonance spectroscopy.
Chiesi’s safety summaries also include other common adverse events such as abdominal pain, diarrhea and vomiting. One company excerpt cited severe adverse reactions in 8 of 29 patients (28 percent) in a specified cohort, without clarifying how that subset relates to the overall 43-child trial.
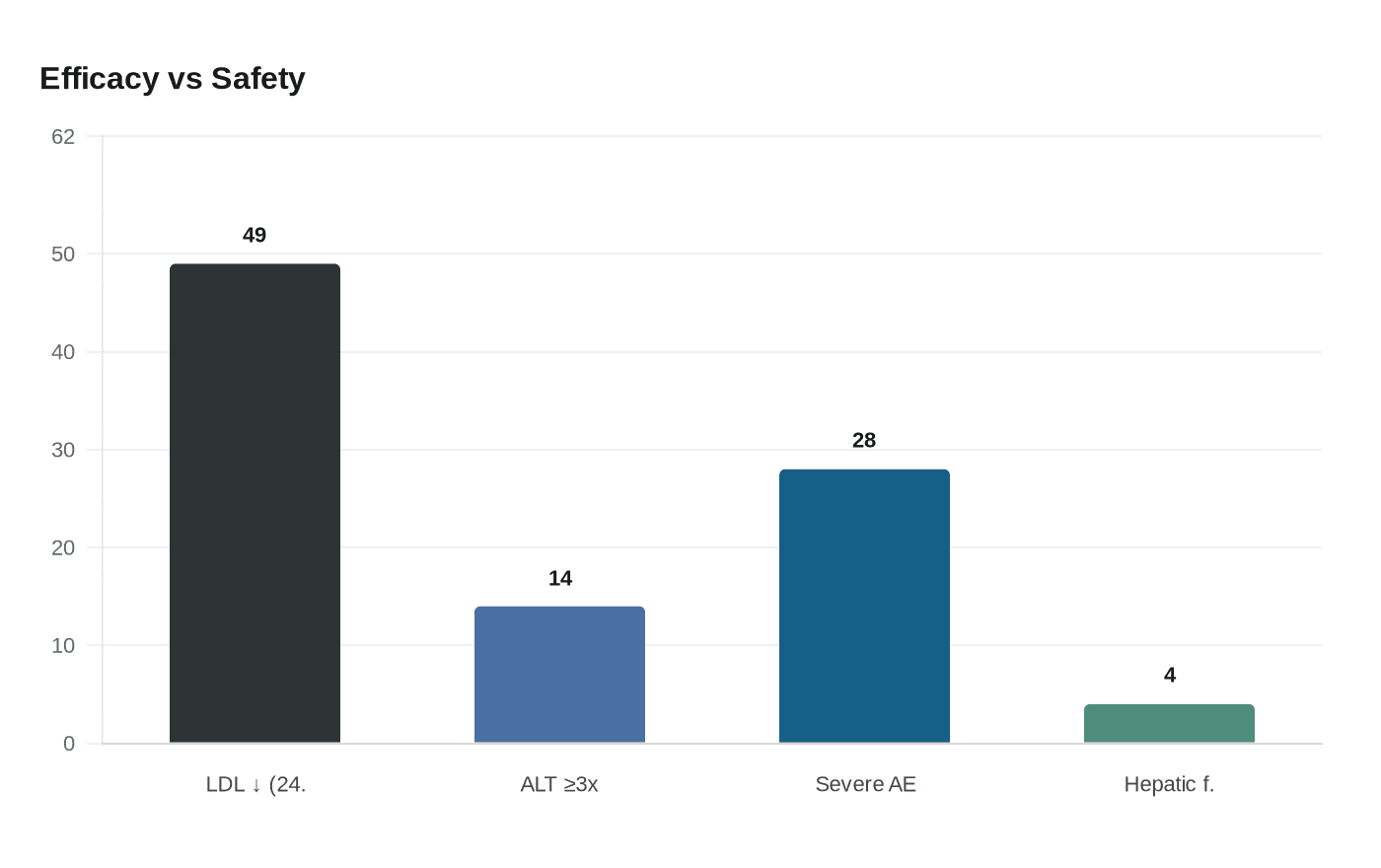
The label clarifies that JUXTAPID is to be used alongside a low-fat diet and other lipid-lowering therapies, including LDL apheresis when applicable. The company noted that in apheresis-treated children mean LDL-C rose gradually over two years, a subgroup trend that will require clinical interpretation.
Clinicians and families now have an approved option to treat very young children with HoFH, a condition that can lead to premature cardiovascular disease. The FDA expansion underscores both the promise of earlier intervention and the need for careful liver monitoring and individualized dosing as patients begin therapy.
Know something we missed? Have a correction or additional information?
Submit a Tip

