FDA grants accelerated approval to HERNEXEOS for HER2‑mutant lung cancer
The FDA on Feb. 26, 2026 approved Boehringer Ingelheim’s HERNEXEOS (zongertinib) for adults with unresectable or metastatic non‑squamous NSCLC with HER2 TKD activating mutations.
The Food and Drug Administration granted accelerated approval on February 26, 2026 to Boehringer Ingelheim’s HERNEXEOS (zongertinib) for adult patients with unresectable or metastatic non-squamous non‑small cell lung cancer whose tumors harbor HER2 (ERBB2) tyrosine kinase domain activating mutations, when detected by an FDA‑authorized test.
The action expands HERNEXEOS into the first‑line setting, a move Boehringer characterizes as the first targeted therapy approved as an initial treatment for adults with HER2‑mutant advanced NSCLC. The company said the approval builds on an earlier accelerated approval in August 2025 for patients who had received prior systemic therapy. The FDA granted Priority Review and Breakthrough Therapy designation to zongertinib and processed the application under the Commissioner’s National Priority Review Voucher pilot program; the agency will post full prescribing information on Drugs@FDA.
The approval is based on data from the Phase Ib Beamion LUNG‑1 trial. Boehringer reported that a treatment‑naïve cohort of 72 patients produced an objective response rate of 76 percent, with 11 percent of patients experiencing a complete response and 65 percent a partial response. The company said 64 percent of responders had a duration of response of at least six months. Those results formed the basis for the first‑line accelerated approval, which the FDA authorized on the strength of objective response rate and duration of response.
Boehringer also cited results from a previously treated cohort that supported the August 2025 approval. Company materials report an objective response rate of 75 percent in 71 patients, with a 95 percent confidence interval of 63 to 83 and a numerator of 53 of 71 responders. The company and the FDA stress these sets of numbers reflect separate cohorts and should not be combined.
Boehringer thanked the trial participants and investigators. “We extend our gratitude to the patients, care partners, and investigators who made this possible. As the first targeted therapy approved as an initial treatment for adults with HER2 (ERBB2)-mutant advanced NSCLC, this is an important step forward for this rare, aggressive cancer,” the company said. Boehringer’s head of U.S. oncology, Vicky Brown, has said “we all have the same goal” of delivering efficient options to patients and has noted HERNEXEOS is the only once‑daily oral therapy cleared for this patient population.
The accelerated pathway carries a regulatory obligation. Boehringer confirmed that continued approval may be contingent on verification and description of clinical benefit in a confirmatory trial, and the company is enrolling patients in that study.
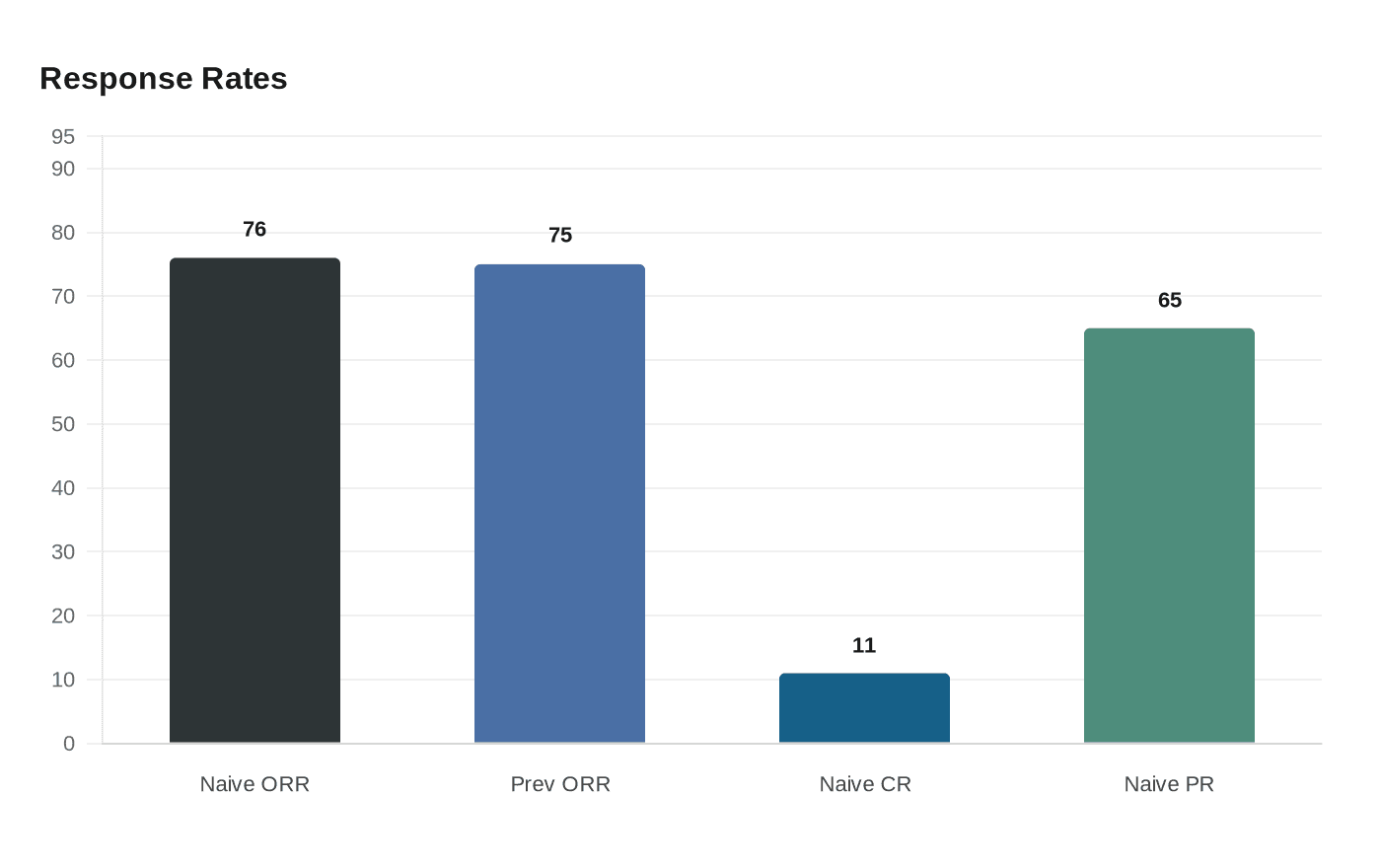
Safety information accompanying the approvals highlights important warnings and precautions including hepatotoxicity, left ventricular dysfunction, interstitial lung disease and pneumonitis, and embryo‑fetal toxicity. Healthcare professionals are asked to report serious adverse events to FDA’s MedWatch at 1‑800‑FDA‑1088. For assistance with single‑patient investigational access, the Oncology Center of Excellence’s Project Facilitate is reachable at 240‑402‑0004 or OncProjectFacilitate@fda.hhs.gov.
The FDA reiterated the decision in a LinkedIn post noting this was the agency’s second approval under the National Priority Voucher pilot program. Additional prescribing details and the full safety profile will be available when the full prescribing information is posted to Drugs@FDA.
Know something we missed? Have a correction or additional information?
Submit a Tip

