Federal probe links growing Salmonella outbreak to moringa supplements
Health agencies are probing moringa leaf supplements after outbreaks sickened dozens across multiple states.

Federal and state health officials have been investigating a widening multistate Salmonella outbreak tied to dietary supplements containing moringa leaf powder, with recent reporting indicating 65 illnesses across 28 states and at least 14 hospitalizations as of late January 2026. The Centers for Disease Control and Prevention and the Food and Drug Administration are working with state partners to trace contaminated products and inform recalls.
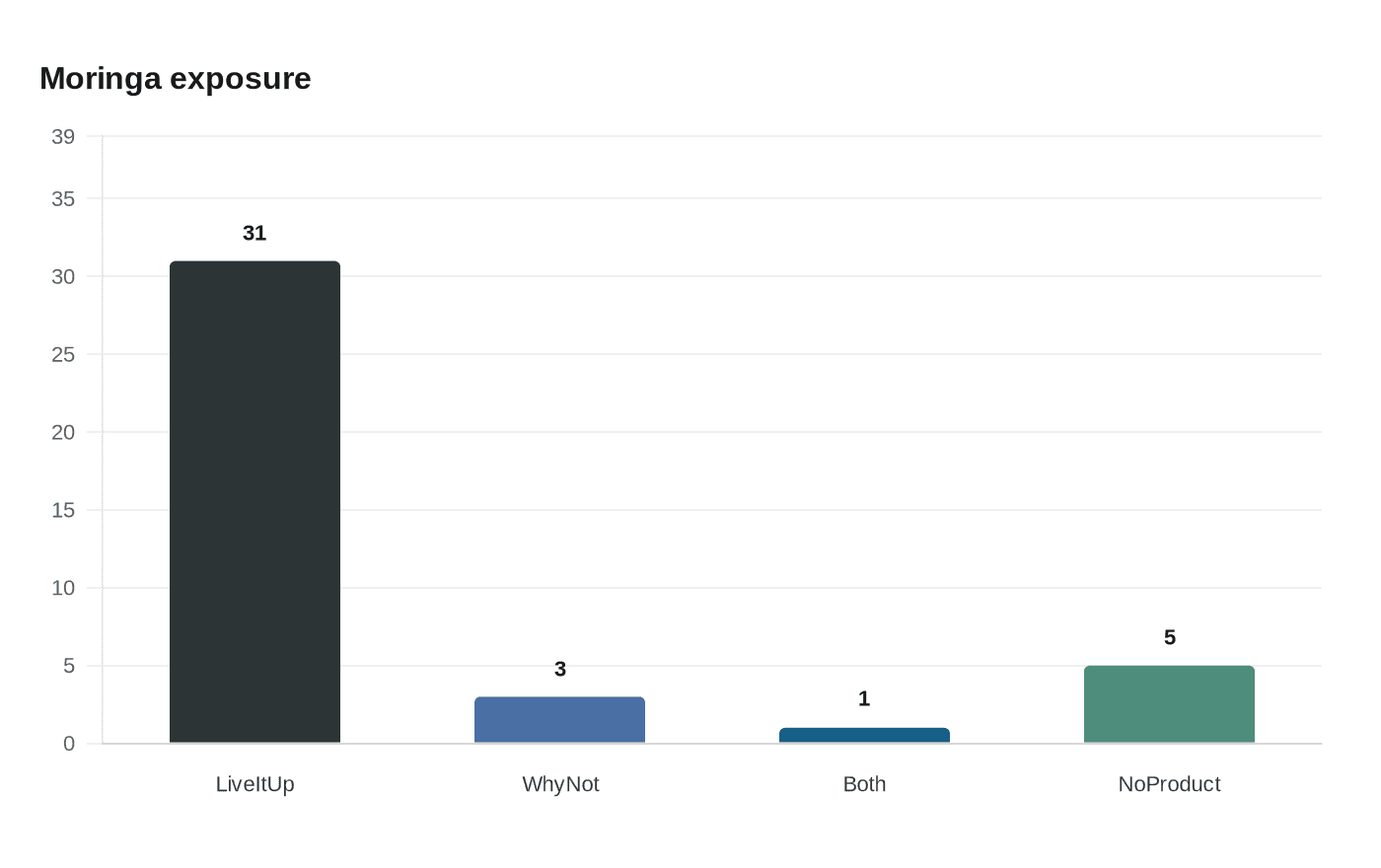
The January 29 update on the larger investigation, cited in media reporting, named two Salmonella serotypes under scrutiny, Typhimurium and Newport, and linked illnesses to powdered supplements and capsules. Interviews conducted with patients show strong product exposure: of 40 people interviewed, 35 reported eating a product containing moringa leaf powder, including 31 who reported Live it Up Super Greens powder only, three who reported Why Not Natural moringa powder capsules only, and one who reported consuming both products, according to FoodSafetyNews. The FDA’s traceback work identified a common manufacturer used by the Live it Up and Why Not Natural products, but the agency has not publicly named that manufacturer.

The newly reported outbreak follows a separate, earlier investigation that federal officials declared over in December 2025. On December 4, 2025, the FDA and CDC said epidemiologic, traceback and laboratory data tied a cluster of Salmonella Richmond infections to a single lot of organic moringa leaf powder supplied to the United States by Vallon Farmdirect PVT LTD of Johdpur, India. That lot was identified as Batch No.: VFD/ORG/MORP/L/24 with a best-by date of November 2027. CDC reported 11 people infected in seven states, with illnesses beginning from May 12 to September 4, 2025; three people were hospitalized and no deaths were reported. Florida officials collected an open container of Food To Live brand moringa leaf powder from an ill person's home, tested it positive for Salmonella, and whole genome sequencing showed the product isolate was closely related to bacteria from sick people, the CDC said.
Regulators have already overseen multiple recalls tied to moringa-containing products. On October 31, 2025, the FDA noted that "As part of this investigation, all Member’s Mark Super Greens dietary supplement powder has been recalled, regardless of lot codes and best by/use before dates." On November 12, 2025, CDC reported that Food To Live of Brooklyn, New York, recalled its Organic Moringa LeafPowder and Organic SuperGreens Powder mix. Federal guidance has been direct: "CDC and FDA advise consumers not to eat and businesses not to sell any of the recalled moringa leaf powder," and consumers are warned that "These products should no longer be available for sale, however they may still be in your home. Check your home for recalled products and throw them away or return them to where you purchased them."
Investigators say laboratory testing, patient interviews and traceback are central to identifying the contamination source and distribution pathways in the supplement supply chain, but public records so far stop short of tying the December Richmond outbreak and the January Typhimurium/Newport outbreak to a single source. Officials have disclosed the Vallon Farmdirect lot in the Richmond case but have not named the manufacturer the FDA identified in the January traceback connecting two branded products. Consumers who have moringa powders or capsules should check recall notices and follow CDC and FDA disposal guidance while the agencies continue their probe.
Know something we missed? Have a correction or additional information?
Submit a Tip

