Galleri blood test fails primary goal in 142,000-patient NHS trial
Grail said the Galleri test did not cut combined stage III and IV cancers in a three-year trial of 142,000 people, though secondary signals and a fall in stage IV disease were reported.

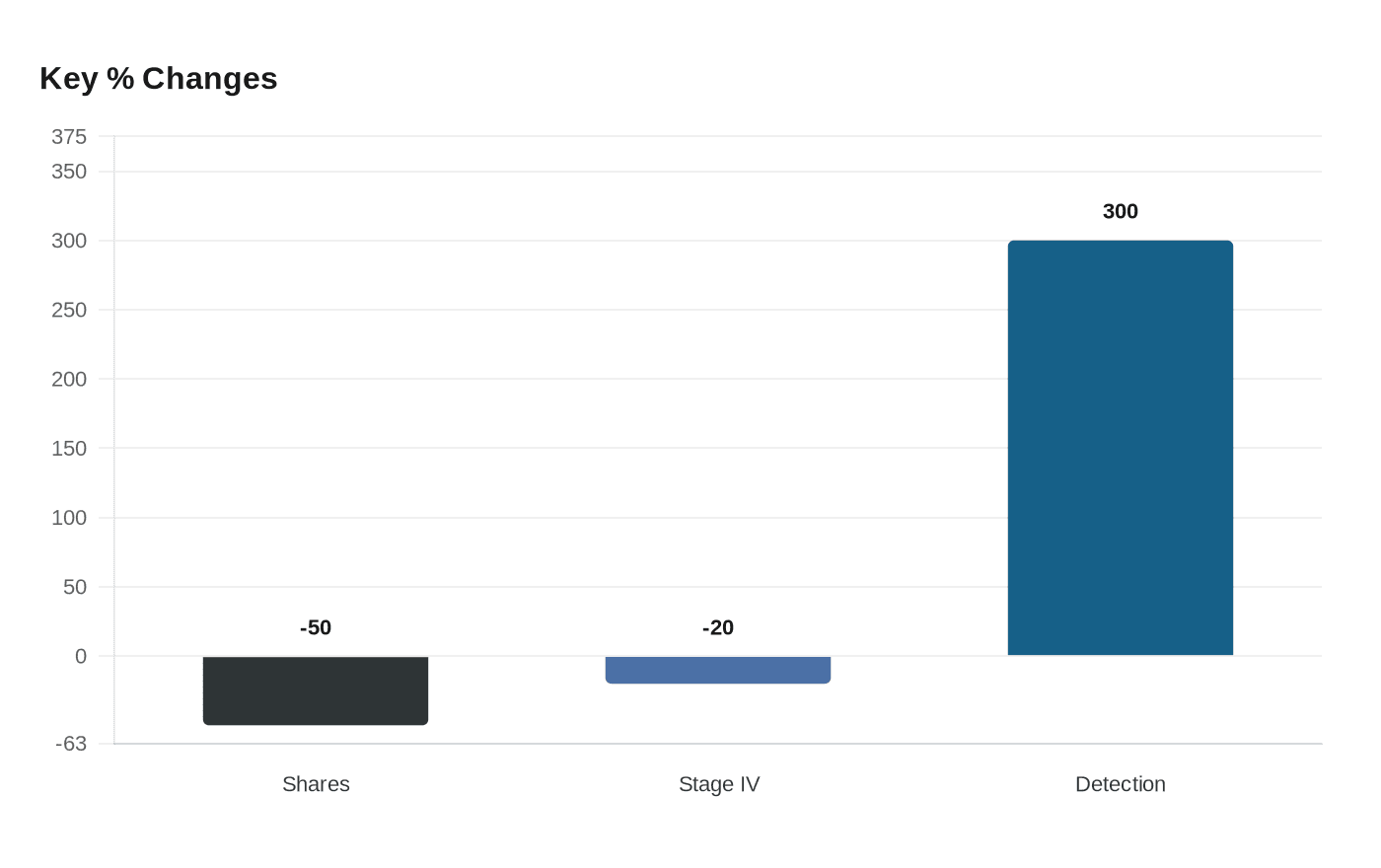
Grail’s Galleri multi-cancer blood test failed to meet the NHS-Galleri trial’s primary objective, the company said, in a setback that sank its shares roughly 50% and raised fresh questions about how molecular screening will be judged by regulators and health systems worldwide. The three-year study enrolled 142,000 asymptomatic participants aged 50 to 77 in England’s National Health Service and aimed to show a statistically significant reduction in combined stage III and IV cancer diagnoses.
“While there was a trend towards reduction in combined stage three and four [cancers], the trial did not meet the primary endpoint,” Grail said in an investor update. Company leaders nonetheless highlighted secondary findings: a separate analysis limited to stage IV cancers showed they “fell by around a fifth,” detection of stage I and II disease rose, and Grail reported a four-fold increase in overall detection rate among screened participants.
Bob Ragusa, Grail’s chief executive, framed the outcome optimistically. “The NHS-Galleri trial provides the strongest evidence to date that multi-cancer early detection can shift the stage at which cancers are detected at a population level,” he said. The company also pointed to a favourable trend over time “in a pre-specified group of 12 deadly cancers,” naming anus, bladder, colorectal, oesophagus, head and neck, liver/bile duct, lung, lymphoma, myeloma/plasma cell neoplasm, ovary, pancreas, and stomach.

Yet independent experts urged caution. Researchers warned that any benefits “remain speculative” until the test is shown to save lives rather than merely change when cancers are found. The NHS said it was looking at the results “carefully.” The distinction between stage shift and mortality benefit is central to debates over population screening, where earlier detection can bring both promise and harms, including overdiagnosis and unnecessary treatment.
Grail has not released the full three-year dataset or the detailed statistical analysis that would show effect sizes, p-values and confidence intervals for the primary and secondary endpoints. The company said it is extending follow-up of the trial by six months to see whether trends firm up to meet statistical thresholds. Grail has also submitted a U.S. premarket approval application for Galleri that includes trial data and plans to publish further results from PATHFINDER 2, a 35,000-subject study, and to continue enrollment in a 50,000-patient REACH study with Medicare.
Analysts and investors reacted sharply. TD Cowen described the failure to meet the primary endpoint as a “major setback,” warning it raises risk to both FDA approval and Medicare coverage. The market move reflected concern that secondary signals may be insufficient for regulators and payers that demand clear proof of clinical benefit.
Technically, Galleri searches for chemical changes in fragments of cell-free DNA that leak from tumours into the bloodstream, with the aim of detecting up to 50 cancer types before symptoms appear. NHS England once hailed the trial at its launch as “the beginning of a revolution.” For now, policymakers and clinicians must weigh an ambiguous set of results: promising signals in subgroups and later-stage falls, but no demonstration yet that routine population screening with this test will reduce deaths.
Know something we missed? Have a correction or additional information?
Submit a Tip