Johns Hopkins Nasal DNA Vaccine Shows Promise Against Tuberculosis in Animals
A nasal DNA vaccine from Johns Hopkins cleared TB bacteria faster and prevented relapse in mice, raising hope for shorter treatment regimens if it reaches human trials.
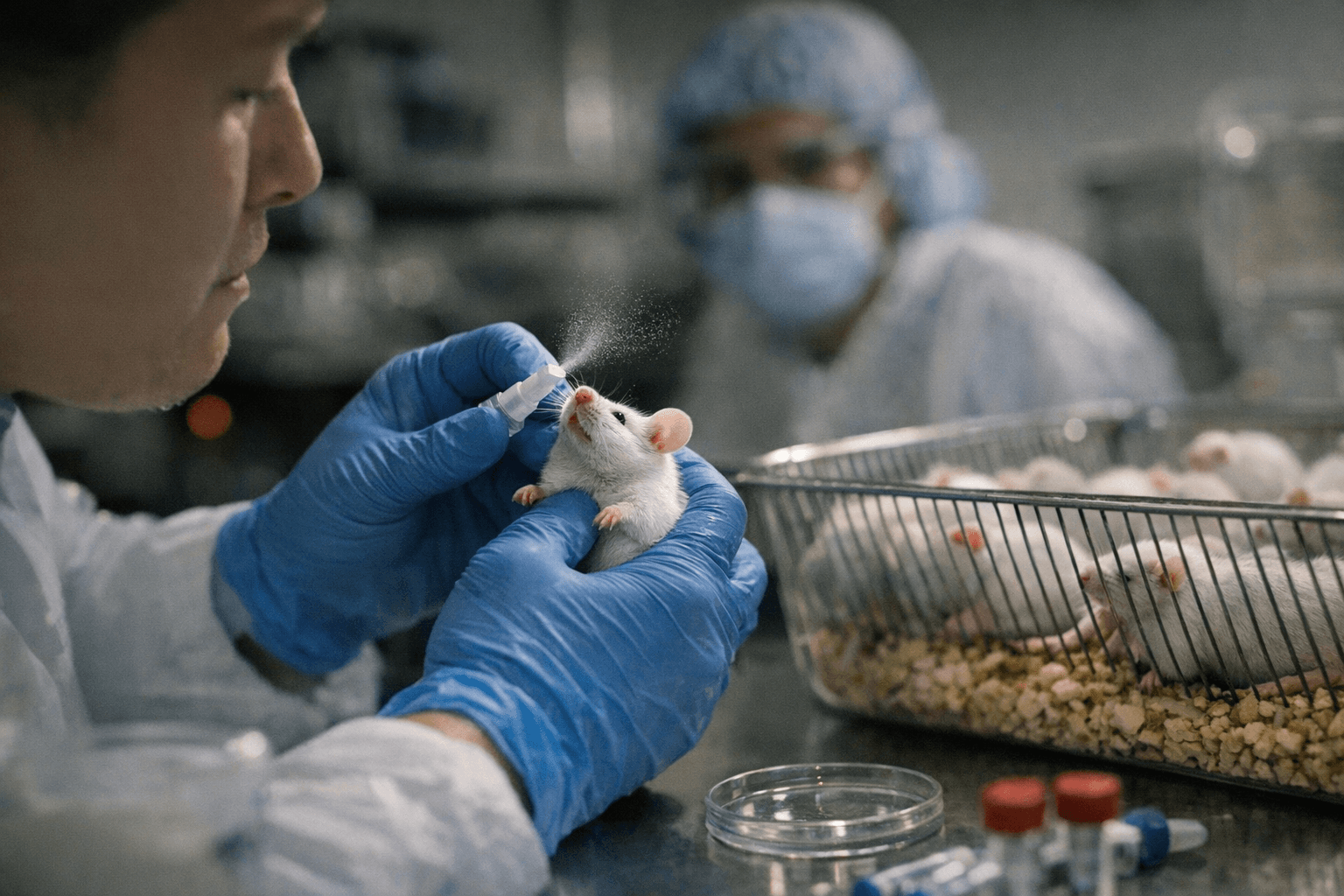
A nose-delivered DNA vaccine developed at Johns Hopkins Medicine helped infected mice clear tuberculosis bacteria faster, reduced lung inflammation, prevented relapse after antibiotics ended, and produced durable immune responses in rhesus macaques — findings that researchers say establish a foundation for eventual human trials in a disease that kills more than a million people every year.
The vaccine was described in the Journal of Clinical Investigation by a research team at Johns Hopkins Medicine and the Johns Hopkins Bloomberg School of Public Health. It fuses two genes with the goal of directing the immune system to fight drug-tolerant bacterial "persisters" — organisms that can survive prolonged antibiotic therapy and contribute to disease relapse.
Lead author Styliani Karanika, M.D., an assistant professor at the Johns Hopkins University School of Medicine, said the formulation fuses rel Mtb and Mip3α and is delivered through the nose to take advantage of specific biological activities in the respiratory tract. The rel Mtb gene is directly implicated in bacterial persistence, while Mip3α recruits immature dendritic cells in the lung to capture and present antigens to the T cells that recognize and fight infection. In mice, the vaccine generated both localized lung and systemic CD4 and CD8 T-cell responses.
"Administered together with first-line TB drug therapy, our intranasal DNA fusion vaccine helped infected mice clear the disease bacteria faster, reduced lung inflammation and prevented relapse after treatment ended," Karanika said.
In rhesus macaques, those immune responses persisted for at least six months, suggesting durability. The primate work, however, measured immune activation only and not response to a TB challenge, and Karanika said more studies are needed before any human clinical trials can be approved. She described the nonhuman primate data as providing "an important translational bridge between the mouse efficacy studies and the additional preclinical work needed before human trials," given how closely primate immune systems resemble those of humans.
The stakes for such research are enormous. Tuberculosis killed 1.23 million people worldwide in 2024, according to the World Health Organization, which also recorded 10.8 million new cases that year. It remains the world's leading cause of death from a single infectious agent. Standard drug therapy runs at least six months, a timeline that strains patient adherence across health systems and creates conditions ripe for resistance. Multidrug-resistant TB carries treatment success rates below 60 percent in many resource-limited settings.
A therapeutic vaccine that shortens that course, even modestly, would carry implications well beyond individual patients. Faster bacterial clearance means less transmission. Reduced relapse rates mean fewer retreatment cycles. And an intranasal delivery method avoids injectable administration, an advantage with real logistical weight in high-burden, low-infrastructure settings across South Asia and sub-Saharan Africa.
TB vaccine development has a long history of promising preclinical candidates that failed in human trials, in part because conventional animal models do not fully replicate how the human immune system responds to infection. The Johns Hopkins team's use of rhesus macaques was intended precisely to narrow that gap, but Karanika and her colleagues have been careful to frame the primate results as a bridge, not a guarantee. Additional preclinical safety and dosing work remains ahead before a first-in-human trial could be submitted for approval, meaning clinical testing is still years away.
Know something we missed? Have a correction or additional information?
Submit a Tip

