Kennedy denies role in FDA rejection of Replimune cancer drug
Kennedy told senators he had nothing to do with Replimune’s FDA rejection, even as the drugmaker faces layoffs, a stock collapse and pressure over its evidence package.

Health Secretary Robert F. Kennedy Jr. drew a bright line between his office and the Food and Drug Administration on Tuesday, telling senators he had nothing to do with the rejection of Replimune Group Inc.’s cancer drug and that the matter rested with FDA Commissioner Marty Makary. The exchange put a political spotlight on a regulatory decision that has already shaken the company, unsettled investors and raised fresh questions about who owns the fallout when a high-stakes cancer treatment falls short of approval.
The FDA issued Replimune a complete response letter on April 10, rejecting RP1 in combination with nivolumab for advanced melanoma. Regulators said the application relied on a single-arm Phase 2 study, RPL-001-16, without a control group, and that the submission needed stronger evidence from a well-controlled trial to show the drug worked. The agency’s redacted letter also said it reviewed additional data from RP1-104, but still concluded the package was not enough to support approval as submitted.

Replimune had spent months preparing for that outcome date. The agency accepted the company’s resubmission on October 20, 2025, and set a PDUFA action date of April 10, 2026. The company said after the rejection that it disagreed with the FDA’s conclusion and believed the agency appeared to contradict positions expressed at a meeting in September 2025. It also said the decision would force it to cut 63 jobs in Woburn, Massachusetts, and scale back U.S. manufacturing.
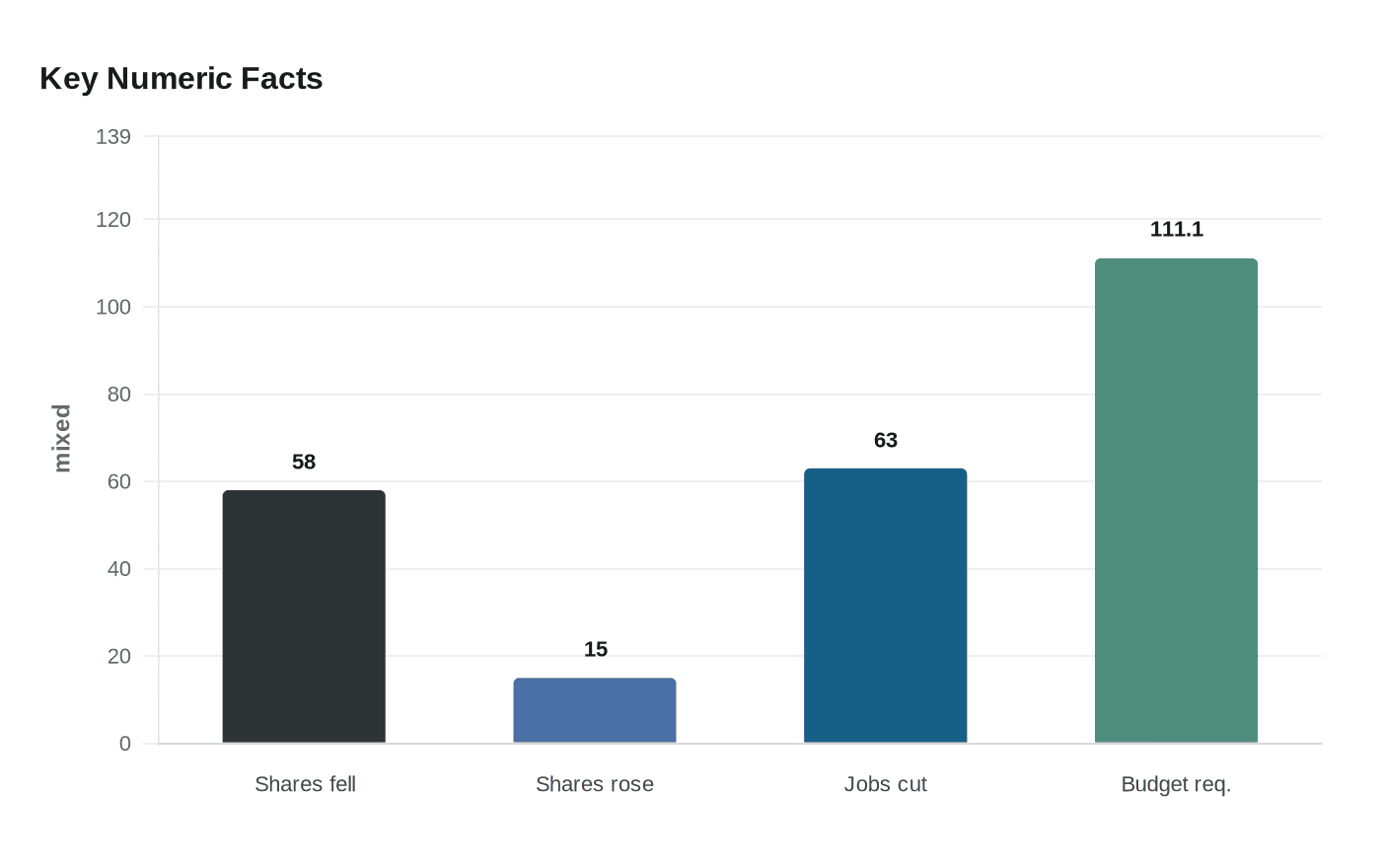
The market reaction was immediate and severe. Bloomberg reported that Replimune shares fell 58% when trading resumed after a halt on April 10, and the stock later sank to fresh lows as analysts questioned the company’s path forward. Shares were still under heavy pressure Tuesday, even after climbing about 15% on the day following a Wall Street Journal opinion piece that said Kennedy’s comments were inaccurate and unfairly shifted blame toward FDA officials.
Kennedy’s testimony came during the Senate Finance Committee’s review of the Department of Health and Human Services’ fiscal 2027 budget request, a $111.1 billion proposal discussed at a 10:00 a.m. hearing in the Dirksen Senate Office Building. The setting underscored a larger fight over the line between scientific review and political accountability. In a system where patients with advanced melanoma are waiting for better options, the FDA is insisting on stronger evidence while lawmakers and advocates press for clarity over whether the ultimate decision belongs to regulators, the secretary who leads them, or both.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

