Neurocrine Biosciences to Acquire Soleno Therapeutics for $2.9 Billion
Neurocrine Biosciences agreed to pay $2.9B for Soleno's VYKAT XR, the only drug approved for Prader-Willi's life-threatening hunger, raising access and pricing questions.

Prader-Willi syndrome leaves patients in a state of chronic, life-threatening hunger with no approved remedy until March 2025. Less than 13 months after that first FDA approval, Neurocrine Biosciences agreed to pay $2.9 billion to take control of VYKAT XR, the drug that filled that void, acquiring Soleno Therapeutics in the largest deal in Neurocrine's history.
The all-cash offer of $53.00 per share, announced April 6, represented a 34% premium to Soleno's prior closing price. Soleno shares surged more than 30% in early trading. The transaction, which is not subject to a financing condition, is expected to close within roughly 90 days pending regulatory clearances and standard closing conditions.
VYKAT XR (diazoxide choline extended-release tablets) is a once-daily oral therapy for hyperphagia in adults and children four years of age and older with Prader-Willi syndrome, a genetic disorder occurring in approximately 1 in every 15,000 live births. Hyperphagia, the hallmark symptom, involves intense, persistent hunger and food preoccupation severe enough to be life-threatening. The FDA granted VYKAT XR breakthrough, fast-track, and orphan drug designations before approving it, signaling both the depth of unmet need and the strength of the clinical data behind it.
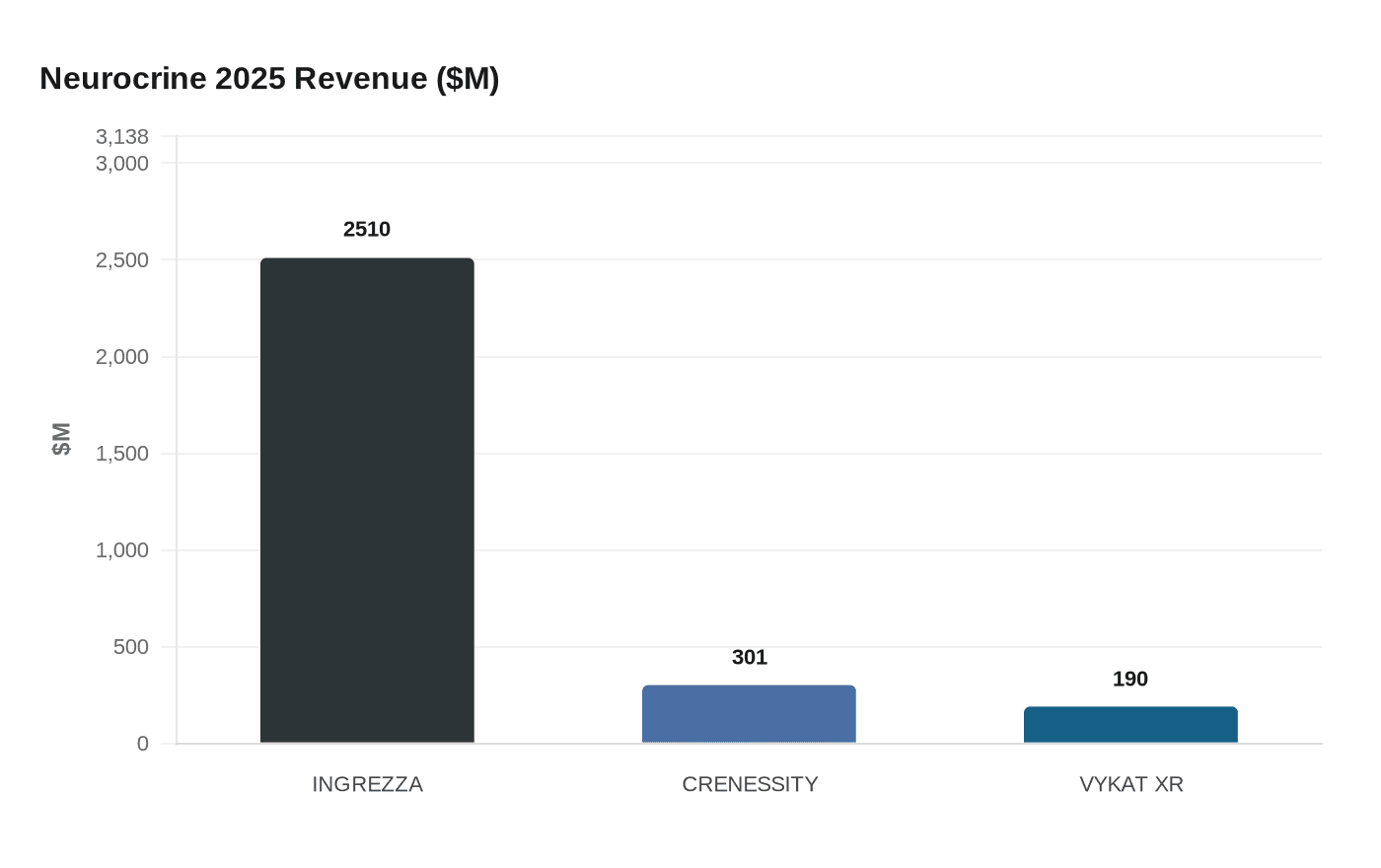
The drug's early commercial performance underpinned Neurocrine's willingness to pay. VYKAT XR generated $190 million in 2025 revenue during its first year on market, including $92 million in the fourth quarter alone. Patent exclusivity is expected to extend into the mid-2040s, giving Neurocrine a multi-decade commercial runway. The company characterized the deal as immediately accretive to 2026 revenue.
The acquisition also reshapes Neurocrine's revenue concentration. INGREZZA (valbenazine), its VMAT2 inhibitor for tardive dyskinesia and Huntington's disease chorea, generated $2.51 billion in 2025, making it an outsized contributor to total revenue. CRENESSITY (crinecerfont), approved for congenital adrenal hyperplasia, added $301 million. VYKAT XR gives Neurocrine a third marketed first-in-class therapy, reducing the portfolio's dependence on any single product and extending its footprint from neuroscience into endocrinology and rare metabolic disorders.
For patients and clinicians treating Prader-Willi syndrome, the central question is whether Neurocrine's commercial scale speeds access or introduces new friction. A larger, better-capitalized company can build broader patient support infrastructure and negotiate with health systems from a position of strength. But payers covering a high-cost orphan drug face a fundamentally different negotiating environment with an established biopharma holding multiple blockbusters than they did with a single-asset rare-disease company. Antitrust review is a standard step in any transaction of this size, and analysts flagged pricing and reimbursement scrutiny as factors to monitor closely given the drug's role in a rare pediatric and adult population with no approved therapeutic alternative.
Soleno's board advised shareholders to view the $53.00 offer as compelling, certain, and immediate value realization. The deal fits a clear 2026 industry pattern: mid-sized biopharma companies acquiring small-company assets with proven clinical differentiation, accelerating sales trajectories, and long patent protection rather than absorbing early-stage pipeline risk. For the Prader-Willi community, the critical variable is whether Neurocrine's commercial infrastructure translates the drug's first-year momentum into broader patient access, or whether a deal that rewarded shareholders handsomely introduces new barriers for the patients VYKAT XR was built to reach.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

