NIH study improves prediction of hypertrophic cardiomyopathy outcomes with new model
A new NIH-supported model may flag hypertrophic cardiomyopathy patients at risk for heart failure, arrhythmias, transplant, and device needs, not just sudden death.
A new NIH-supported model is designed to help cardiologists identify which hypertrophic cardiomyopathy patients need closer surveillance sooner, shifting risk assessment beyond sudden cardiac death alone. The advance matters because HCM, a chronic heart condition that affects about 1 in 500 people, can stay quiet for years while still carrying the risk of serious cardiac events.
The National Institutes of Health said Monday, May 11, 2026, that the study drew on the Hypertrophic Cardiomyopathy Registry, a prospective, NHLBI-funded cohort built to improve prognostication by combining cardiac MRI, biomarkers and genetic data. The registry enrolled nearly 2,700 patients at 44 HCM-specialized sites across North America and Europe, with earlier descriptions placing the broader cohort at 2,755 patients across 44 sites in 6 countries. Researchers followed patients for an average of seven years, giving the model enough longitudinal data to test whether more detailed information could predict outcomes more accurately.
That broader lens is the key shift. Christopher Kramer, a principal investigator and cardiologist at the University of Virginia, said current guidelines are imperfect because they focus mainly on sudden cardiac death and do not adequately predict heart failure or other fatal and nonfatal cardiac events. The new model tried to fill that gap by looking at heart failure, nonfatal arrhythmias, device implantation and transplant needs, the kinds of decisions that can change follow-up intensity, medication plans and when to move a patient toward an ICD or specialty care.
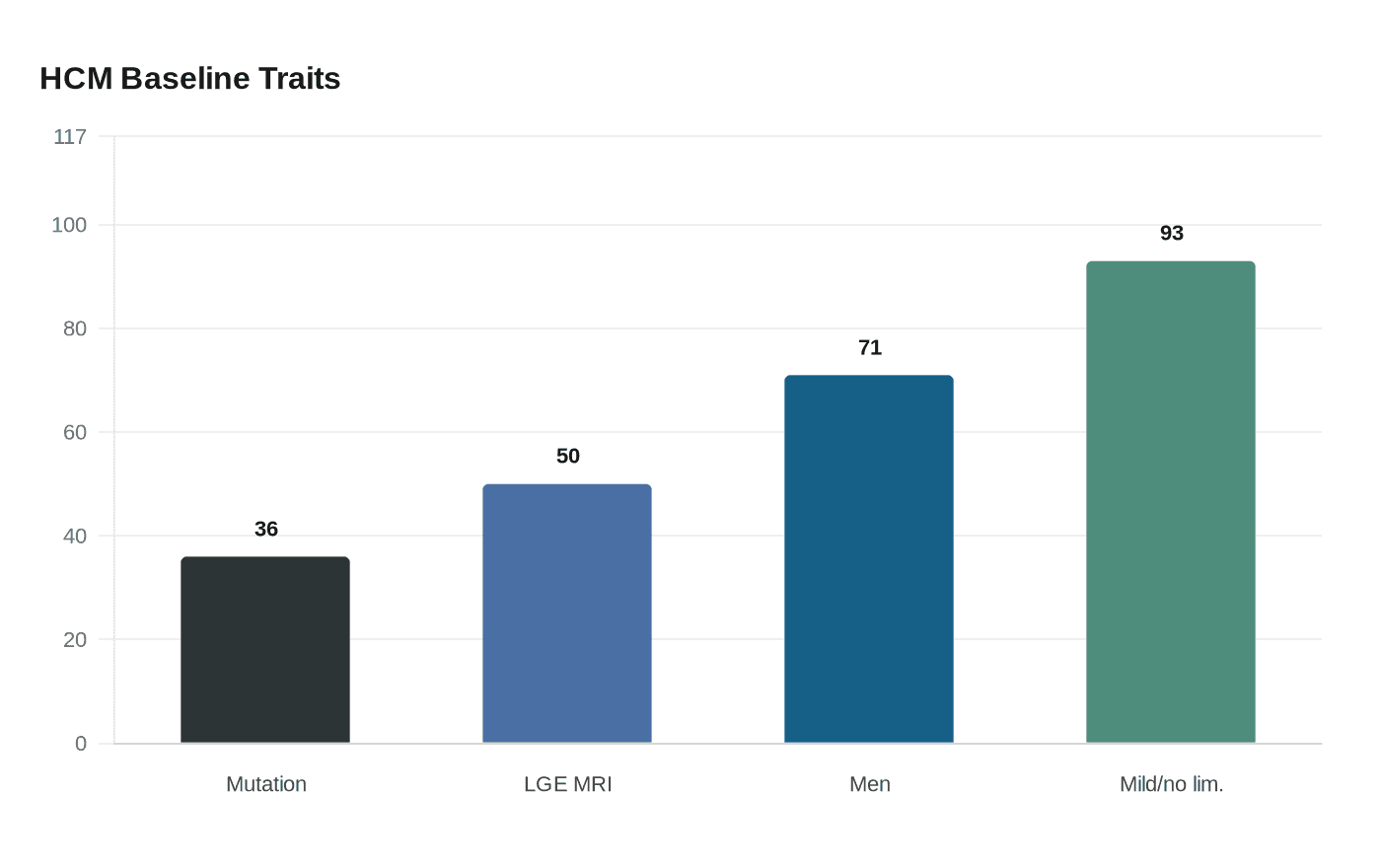
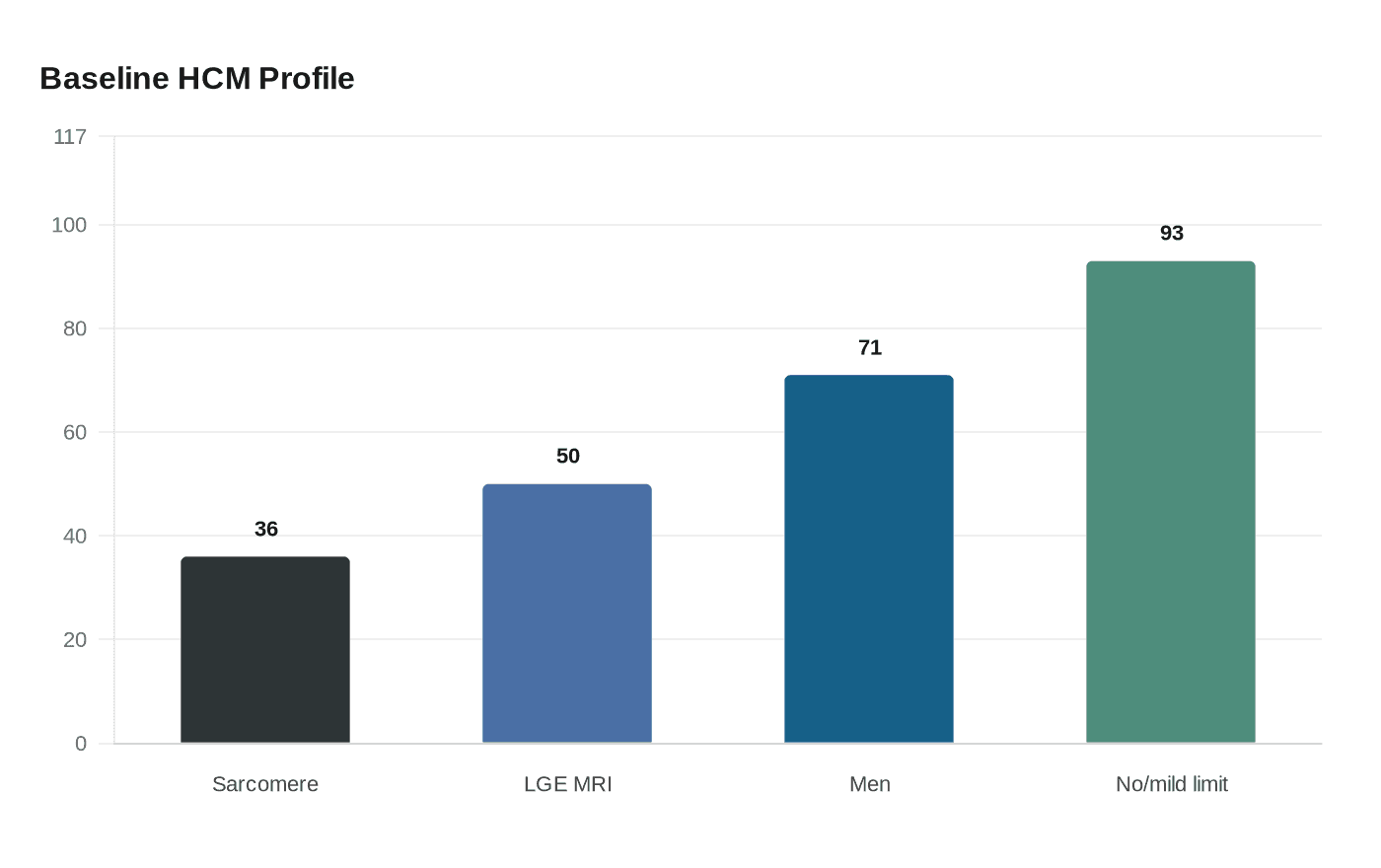
The study found that several features were linked with worse outcomes, including scarring on imaging, weight, impaired heart muscle function, prior heart failure and elevated NTproBNP levels. Prior HCMR baseline data showed that 36% of participants had a sarcomere mutation, 50% had late gadolinium enhancement on MRI, the mean age was 49, 71% were men and 93% had no or only mild functional limitation at baseline. That profile underscores why more precise tools are needed: many patients appear relatively low-risk at first glance, yet still require better triage over time.
The American Heart Association has said HCM is the most common inherited heart disease and that it can lead to atrial fibrillation, stroke, heart failure and worse quality of life. It also notes that its current sudden cardiac death calculator applies to patients age 16 and older and excludes several groups, including elite athletes, metabolic or syndromic HCM, and secondary-prevention ICD cases. For cardiologists, the new model points toward more personalized monitoring now, while the bigger promise lies in deciding earlier who needs intervention and who can be watched less aggressively.
Know something we missed? Have a correction or additional information?
Submit a Tip