NIH study maps pancreatic cells, revealing clues to diabetes risk
A map of islet cells from 299 non-diabetic donors shows how pancreas cell mix varies by person, a clue to why diabetes risk is not the same for everyone.

A federally funded map of human pancreatic islet cells is giving diabetes researchers a more detailed picture of why blood sugar control varies so much from person to person. By analyzing islets from 299 organ donors without diabetes, scientists identified wide differences in the balance of hormone-producing cells that help regulate glucose, a finding that could sharpen risk prediction and point drug development toward more precise targets.
The study, published in Nature Communications under the title “Heterogeneous endocrine cell composition defines human islet hormone secretion traits,” focused on a healthy-donor reference set rather than a diabetes cohort. That matters because pancreatic islets are the clusters of cells that manage blood sugar by releasing hormones such as insulin from beta cells, glucagon from alpha cells, and somatostatin from delta cells. Looking across 299 donors gave researchers a scale large enough to see patterns that smaller studies would likely miss.
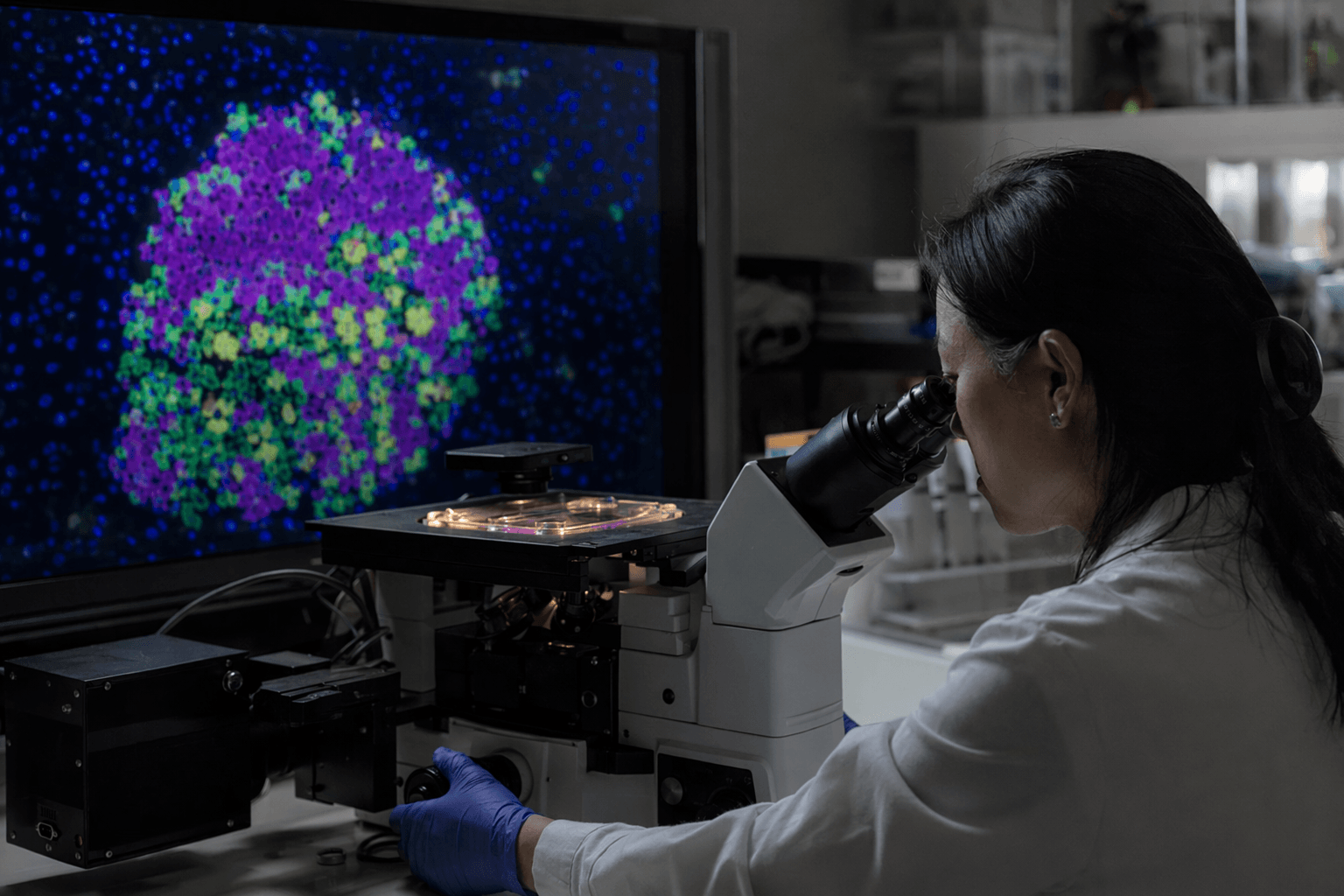
The work drew on the Integrated Islet Distribution Program, funded by the National Institute of Diabetes and Digestive and Kidney Diseases and led by City of Hope in Duarte, California, since 2002. City of Hope processes and distributes human islets from cadaveric donors for approved investigators, and the program has become a major national research engine, supporting nearly 200 investigator-initiated projects in its last project period and producing 168 publications in the first four years of the previous cycle. For diabetes science, those tissues are unusually valuable: NIDDK describes human islets as essential for understanding islet cell biology and building new therapies.
The practical significance is not that the study treats diabetes today, but that it may help explain why some people are more vulnerable than others long before symptoms appear. If future research can link a donor’s cellular mix, molecular signature or inherited traits to how resilient their islets are, clinicians may eventually move beyond broad labels and toward more individualized risk estimates. That could improve early detection, prevention strategies and the search for therapies tailored to a patient’s biology rather than a one-size-fits-all category.
The scale of the new map also raises the bar for the field. A prior reference map used 192,203 cells from 65 donors, far smaller than the 299-donor dataset. NIDDK says decades of islet research recently helped lead to the first FDA approval of islet transplantation for some adults with difficult-to-manage type 1 diabetes, a reminder that donor-islet science can eventually reach the clinic. This latest map stays in the basic-science stage, but it expands the biological atlas that future diabetes tests and treatments will likely depend on.
Know something we missed? Have a correction or additional information?
Submit a Tip

