One in eight U.S. adults now takes GLP-1 drugs as use broadens
One in eight U.S. adults now uses a GLP-1 drug, and the boom is colliding with obesity's scale, high prices and a still-shaky market for compounded versions.

One in eight U.S. adults is now taking a GLP-1 drug, a sign that medicines once framed mainly as weight-loss shots have moved into routine care for obesity, diabetes and other chronic conditions. KFF’s November 2025 poll found that 12% of adults said they were currently using one of the drugs, such as Ozempic or Wegovy.
That level of use lands in a country where obesity remains widespread. The Centers for Disease Control and Prevention says 41.9% of U.S. adults had obesity in 2017 to March 2020, up from 30.5% in 1999 to 2000, and more than 100 million adults have obesity. GLP-1s are now part of that chronic disease picture, not just a culture-war symbol about body size and dieting.
The medical case has broadened as well. On March 8, 2024, the Food and Drug Administration approved Wegovy to reduce the risk of cardiovascular death, heart attack and stroke in adults with established cardiovascular disease and either obesity or overweight. In 2024, the agency also approved Zepbound to treat moderate to severe obstructive sleep apnea in adults with obesity. That expansion helps explain why the drugs now sit at the center of debates over chronic disease, food culture, affordability, access and long-term dependence.

But the market around GLP-1s has grown faster than the guardrails around it. After months of pressure on tirzepatide supply, the FDA said on April 1, 2026, that the national shortage had been resolved and that GLP-1 supply was beginning to stabilize. The shortage had already pushed many consumers toward compounded versions and other workarounds, a reminder that scarcity creates a fast-moving gray market where oversight is weakest.
Price remains the sharpest pressure point. KFF found in 2024 that about half of adults who had taken GLP-1 drugs said they had trouble affording them even with insurance. As more patients qualify under clinical guidance, that cost burden is likely to shape who stays on treatment and who drifts back to older, less effective options.
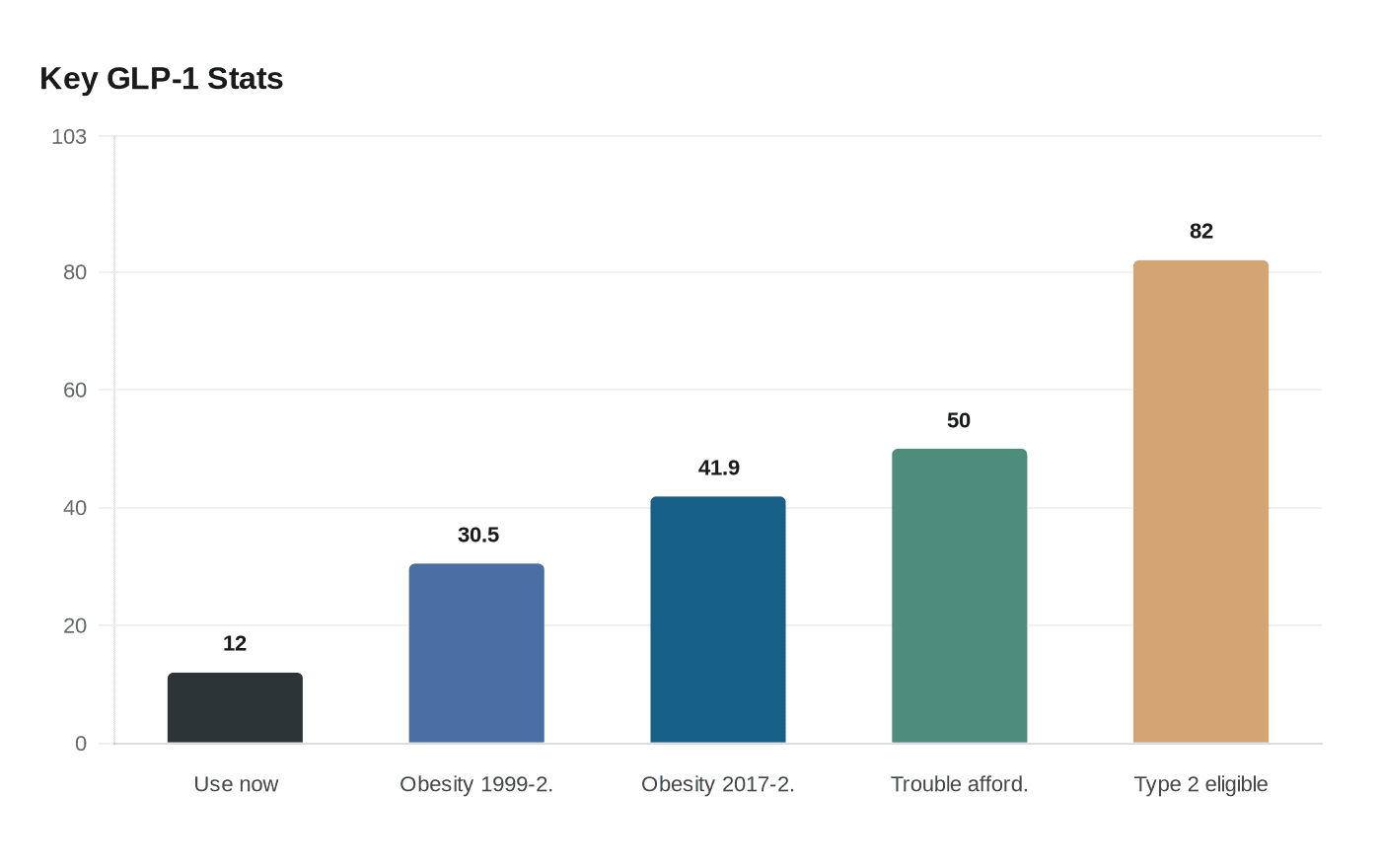
The eligible population is large. CDC estimated that 82% of U.S. adults with type 2 diabetes may be eligible for GLP-1, SGLT-2 or both under updated ADA and EASD guidance. That helps explain why GLP-1s have become more than a pharmaceutical trend. They are now a test of whether the health system can pay for, regulate and sustain a therapy that is moving from novelty to mainstream chronic care.
Know something we missed? Have a correction or additional information?
Submit a Tip

