Patients with deadly cancer push for unapproved drug access through FDA path
Patients with advanced pancreatic cancer are pressing for drugs the FDA has not fully approved. The access path offers hope, but not proof the medicine works.
Patients facing one of the deadliest cancers are driving the debate over unapproved treatment because time is short and standard options are thin. The Food and Drug Administration’s expanded-access pathway, often called compassionate use, is meant for people with serious or immediately life-threatening disease who have no comparable or satisfactory alternative therapy. It can open a door to an investigational drug, but it also comes with a hard warning: the medicine is not approved for that use, may not help, and can trigger unexpected serious side effects.
The FDA has said it has allowed expanded access to experimental drugs and biologics since the 1970s, and in oncology it launched Project Facilitate on May 31, 2019, to help providers navigate requests. Even with that support, the path still depends on three approvals, from the treating physician, the manufacturer supplying the drug, and the FDA. The agency also says expanded use cannot interfere with clinical investigations that could support marketing approval, and patients may face costs that insurance or Medicare do not cover. For people without a trial slot, without a willing company, or without coverage for extra expenses, the promise of access can still end in a wait.
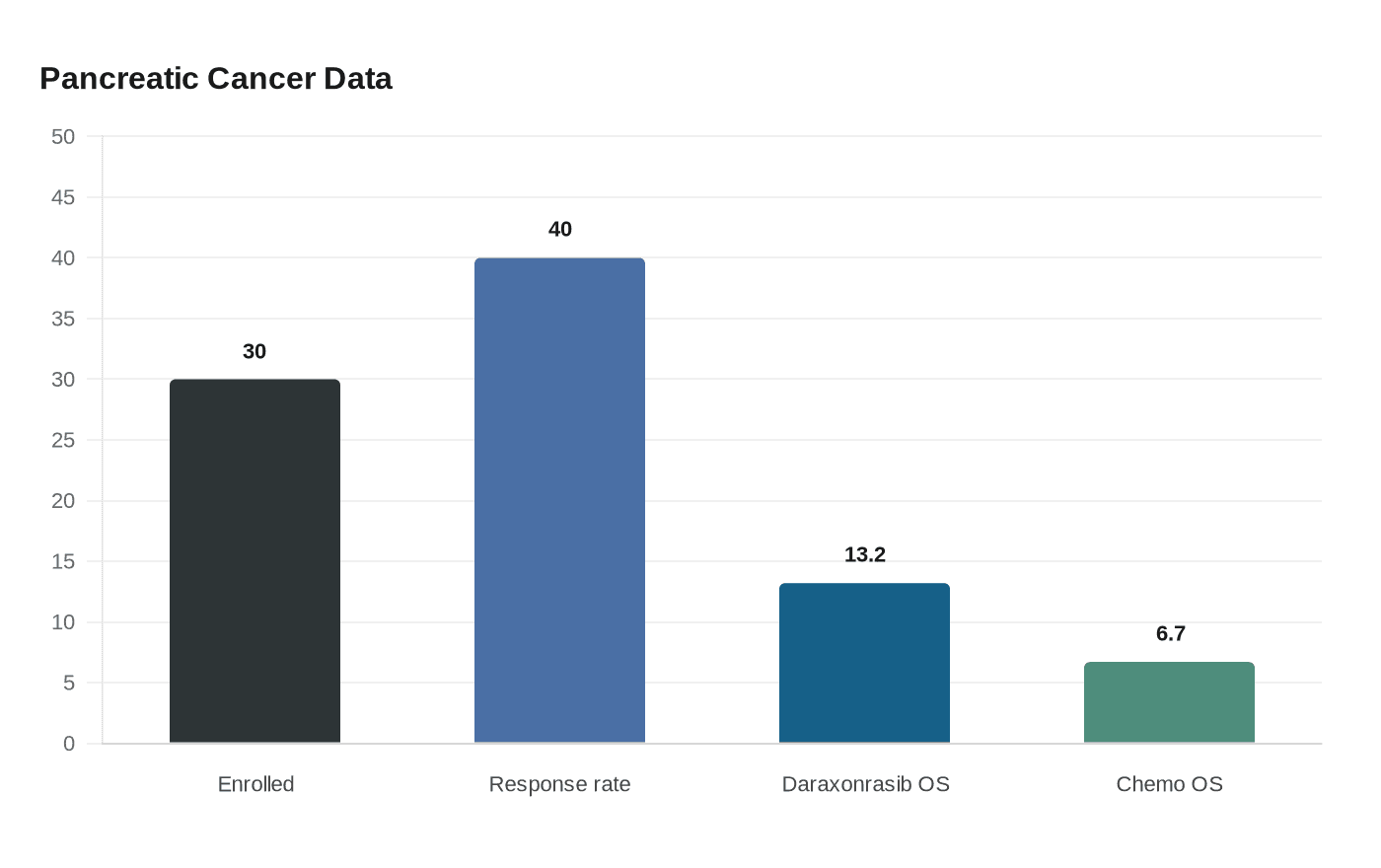
That tension has sharpened around pancreatic cancer. On December 4, 2024, the FDA granted accelerated approval to zenocutuzumab-zbco, sold as Bizengri, for advanced pancreatic adenocarcinoma with an NRG1 gene fusion. The agency described it as the first FDA approval of a systemic therapy for pancreatic adenocarcinoma with that biomarker. The pivotal eNRGy study enrolled 30 adults with advanced or metastatic NRG1 fusion-positive pancreatic adenocarcinoma, and FDA reported a 40% objective response rate in that group. Even so, accelerated approval is not the same as a full proof of benefit, and the cancer-drug framework still relies on postmarketing trials to verify clinical value.
Recent data on daraxonrasib have only intensified the pressure. On April 13, 2026, Revolution Medicines announced positive topline phase 3 results in metastatic pancreatic ductal adenocarcinoma, saying the trial met its primary and key secondary endpoints and that it planned to seek FDA review in a future application. Coverage of the readout reported median overall survival of 13.2 months with daraxonrasib versus 6.7 months with chemotherapy, and PanCAN called the results a meaningful advance on April 14, 2026.
For patients and families confronting a fast-killing disease, those signals are reason enough to push for access before full approval. For regulators, they are a reminder that early access can offer time, but not certainty, and that the people left waiting are often those who cannot get a trial, a company sponsor, or the money to make the pathway work.
Know something we missed? Have a correction or additional information?
Submit a Tip

