Science Corporation prepares first U.S. trials of biohybrid brain interface
Science Corporation is preparing to place a first sensor in a human brain, testing whether a biohybrid interface can heal tissue instead of just reading it.

Science Corporation is preparing its first U.S. human trials of a biohybrid brain-computer interface, a step that would move the startup from promise to surgery as it tries to place the first sensor of a future system into a patient’s brain. The device is intended to do more than transmit signals. Science says the long-term goal is to combine lab-grown neurons with electronics and use gentle electrical stimulation to help damaged brain or spinal cord cells recover.
The first test will be a threshold moment for a field that has moved fast in headlines but slowly in medicine. If the trial succeeds, it would show that a more organic interface can be implanted safely and function in living human tissue. If it fails, it will reinforce how difficult it remains to make brain hardware durable, precise and gentle enough to avoid harming the tissue it is meant to serve.
Science was founded in April 2021 by Max Hodak, the former Neuralink president and co-founder, and has been building a broader portfolio around neural repair and sight restoration. The company brought on Murat Günel, chair of Yale Medical School’s Department of Neurosurgery, as a scientific adviser after two years of discussions. Günel’s role centers on the surgical placement of the first sensor for the future interface.
The company has already shown it can turn acquired technology into a regulated medical product. Science bought the PRIMA vision-restoring implant in 2024 and has since pushed it through clinical testing for people with advanced macular degeneration and similar conditions. In a 2025 study, Science said PRIMA was tested in 38 patients at 17 sites across five countries, with 80% showing meaningful improvement in visual acuity and 84% saying they could read letters, numbers and words. Science says PRIMA received U.S. Food and Drug Administration Breakthrough Device status in 2023 and is moving through approval efforts in the United States and Europe.
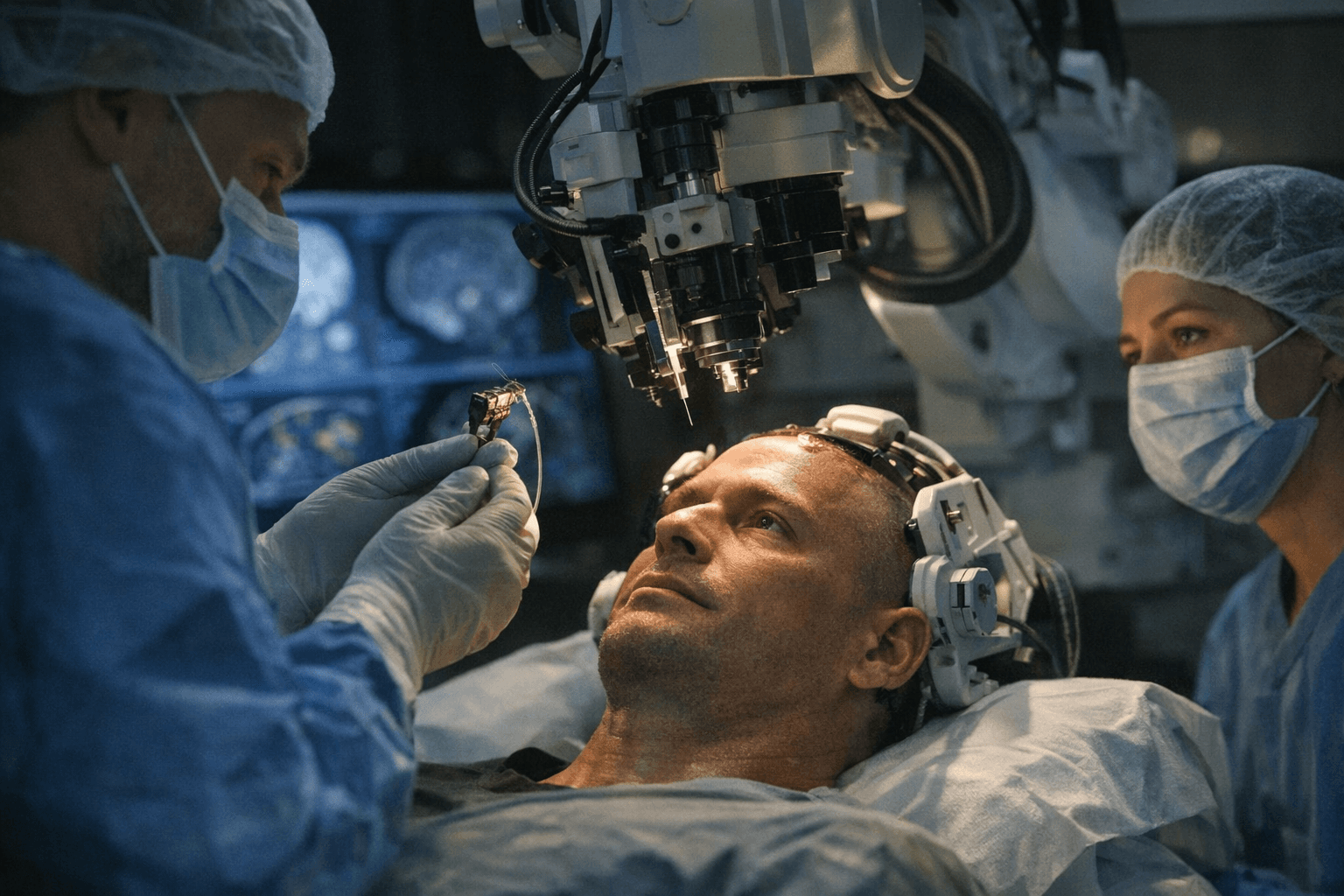
That track record matters because the brain-interface project sits at the intersection of high medical need and high uncertainty. Existing implanted sensors from companies such as Neuralink have already helped some patients with ALS, spinal cord injuries and related conditions control computers or produce text by thought, but the eligible patient pool is still relatively small and regulatory hurdles remain high. Science argues that traditional metal probes and electrodes can injure brain tissue over time, which is why it is pursuing a more biological approach.
The distinction from Neuralink is also one of purpose and pace. Neuralink’s PRIME study is an early feasibility trial for people with tetraparesis or tetraplegia, began on January 9, 2024, and has an estimated completion date of January 2031. Science’s effort is narrower at the start but broader in its ambition, aiming not only at communication for paralysis but also at treatment, healing and, eventually, new sensory capabilities. The company’s $230 million Series C round in March 2026, reported at a $1.5 billion valuation, gave that bet more capital, but the real test will begin in the operating room.
This article was produced by Prism’s automated news system from verified source data, official records, and press releases, then run through automated quality and moderation checks before publishing. The system is built and supervised by the people who set the standards it runs under. Read our full AI policy.
Know something we missed? Have a correction or additional information?
Submit a Tip