Science Translational Medicine paper reports levetiracetam cuts amyloid buildup in models
Researchers report levetiracetam reduced amyloid‑beta accumulation in animal models and human neurons, raising hopes for repurposing but underscoring need for clinical trials.
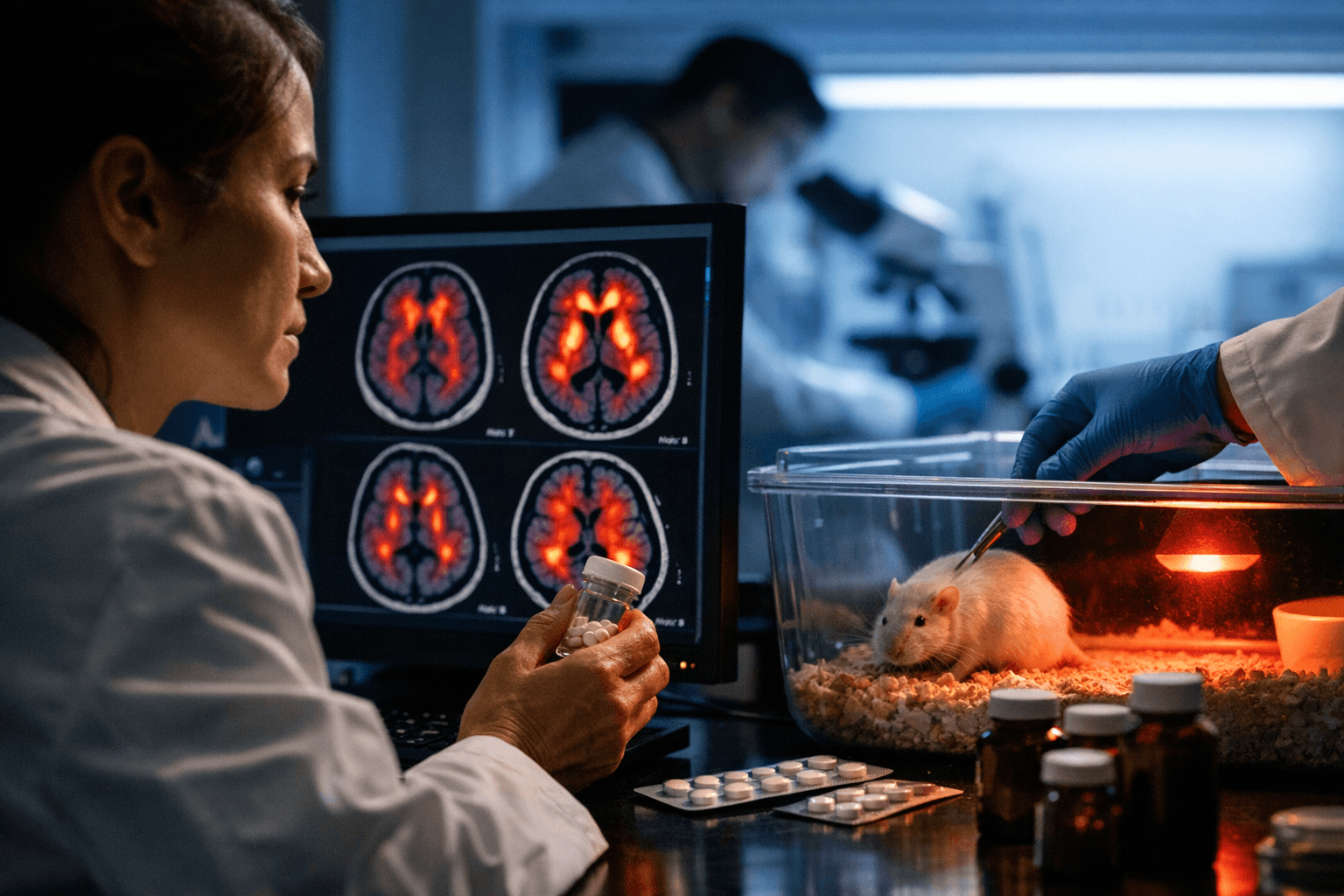
Researchers publishing in Science Translational Medicine reported that levetiracetam, an FDA‑approved anticonvulsant, reduced the buildup of Alzheimer’s‑related amyloid‑beta in animal models and human neurons and preserved synapses in engineered mice, a result that could change prevention research as the number of Americans with Alzheimer’s is projected to reach 14 million by 2060.
The study, described in media summaries of the peer‑reviewed paper, used a three‑pronged approach: engineered mouse models predisposed to amyloid accumulation, laboratory human neurons, and postmortem brain tissue from people described as at high risk of developing Alzheimer’s. Coverage of the paper emphasizes biochemical and tissue‑level reductions in amyloid‑beta and fewer disrupted synapses in mice, but stops short of clinical claims. "The precise mechanism by which levetiracetam reduces amyloid‑beta buildup is unclear," the coverage states, and authors caution that substantial translational work is required before considering human prevention trials.
Levetiracetam is long established in neurology. Its chemical name is levetiracetam ((S)-α-ethyl-2-oxo-1-pyrrolidine acetamide), and it is approved by the FDA for oral adjunctive therapy for focal, myoclonic, and primary generalized seizures since 2000, with intravenous approval in 2006 for patients older than 15 when oral dosing is not feasible. As background for potential repurposing, pharmacology summaries note a well‑characterized mechanism of action: "As a broad-spectrum AED, levetiracetam ((S)-α-ethyl-2-oxo-1-pyrrolidine acetamide) has unique mechanisms of action that differs from other AEDs. A study published recently revealed that through binding, levetiracetam modulates the activity of synaptic vesicle protein 2A in brain neurons to maintain a normal level, and as a result seizures reduce." Absorption is rapid and nearly complete with peak serum concentrations reached in roughly 0.6–1.3 hours; elimination half‑life averages about 6–8 hours in young adults and 10–11 hours in older patients.
The drug’s existing clinical record matters for public health planning. Systematic reviews and randomized trials establish levetiracetam as an effective antiepileptic with a tolerability profile that has made it attractive for older adults and women of childbearing potential. The World Health Organization commentary cited in expert summaries concluded that "Levetiracetam has specific advantages above current EML anti‑seizure medications, including few drug‑drug interactions, no long‑term side effects, good tolerability in older people and the safest teratogenic profile of all anti‑seizure medications (ASMs;)." In pediatric emergency care, a National Institute for Health Research summary of a trial concluded: "Levetiracetam is as effective as phenytoin at stopping prolonged epileptic seizures in children." The study found that "Levetiracetam stopped 70% of children convulsing compared with 64% for phenytoin within 35 to 45 minutes." "Adverse events were similar," the report adds.
For clinicians, patients, and policymakers the headline is both hopeful and cautionary. Repurposing an inexpensive, widely available generic medicine could accelerate equitable access to new dementia prevention approaches if clinical benefit is proven. But the Science Translational Medicine results remain preclinical: no human interventional data demonstrating prevention of cognitive decline are reported in the summaries, and key translational details such as doses used in models and characteristics of human tissue samples must be reviewed in the full paper. The next necessary steps are rigorously designed clinical trials and transparent reporting of methods so regulators, health systems, and communities can weigh efficacy, safety, and equitable deployment.
Know something we missed? Have a correction or additional information?
Submit a Tip

