Shared Biology of Autism and Alzheimer's May Transform Treatment for Both
Autistic adults are diagnosed with Alzheimer's at rates up to eight times higher than the general population, and the shared biology behind both conditions may unlock new treatments for each.

Two conditions long treated as opposites, one beginning in early childhood, the other arriving late in life, are revealing a surprisingly shared molecular architecture. The growing body of research connecting autism spectrum disorder (ASD) and Alzheimer's disease (AD) is not just an academic curiosity; it carries direct implications for how clinicians screen patients, how drug developers target therapies, and how millions of families plan for the future.
A Statistical Signal Too Large to Ignore
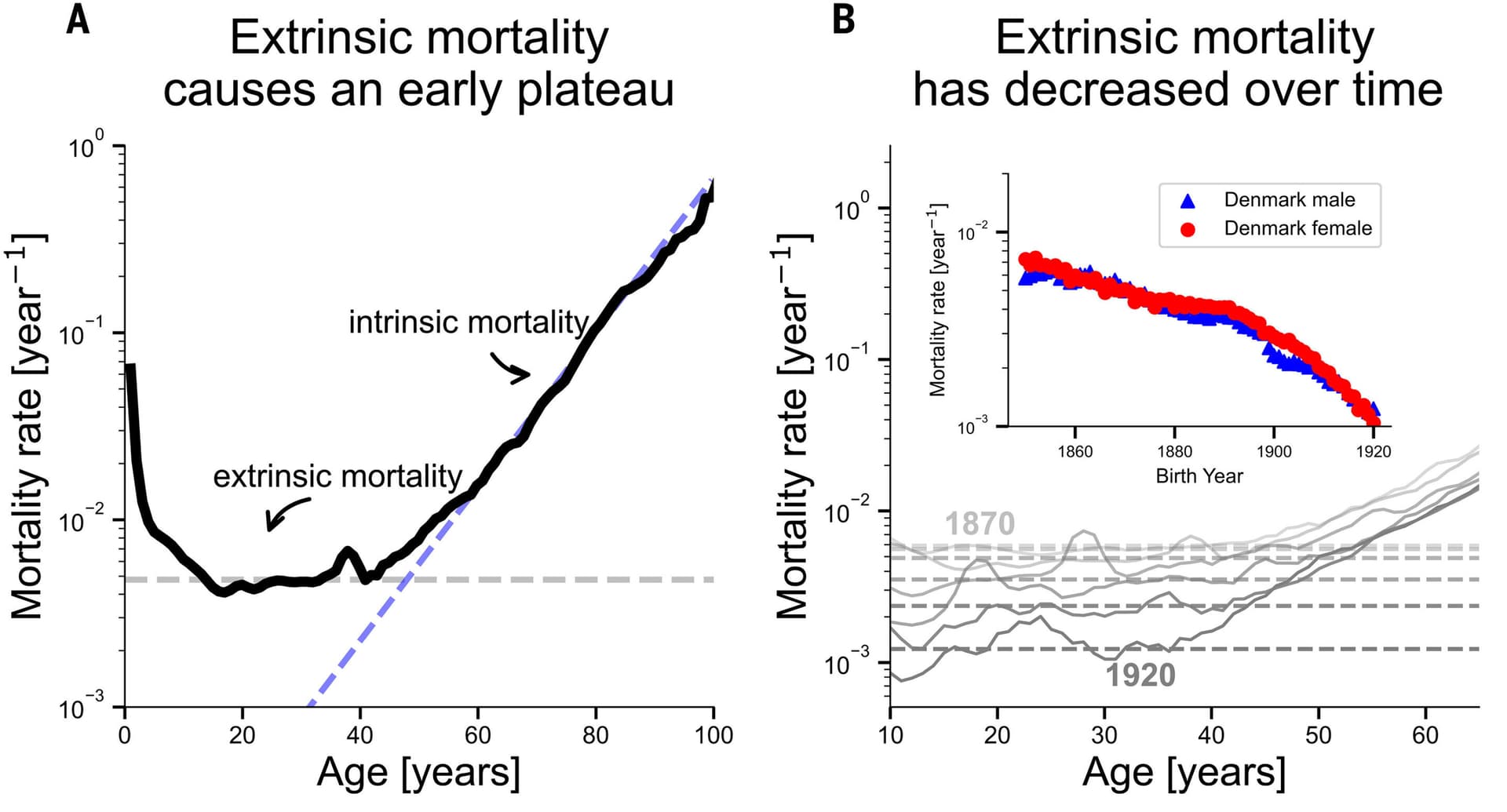
The epidemiological picture arrived with force in a landmark study published in *Autism Research* in April 2025. Researchers from Boston University School of Public Health analyzed longitudinal Medicare and Medicaid data covering more than 114,000 autistic adults aged 30 and older between 2011 and 2019. The study found that 8% of autistic adults without intellectual disabilities and 8.9% of those with intellectual disabilities had been diagnosed with dementia, rates far higher than the roughly 1% prevalence seen in the general population. Perhaps more striking was the timing: mean age at Alzheimer's disease and related dementia onset among autistic adults was 59.3 years, arriving years or even decades before typical Alzheimer's diagnoses in the broader population.
Earlier research using Medicaid claims data had already established a troubling pattern. Autistic individuals were found to be approximately 2.6 times more likely to be diagnosed with early-onset Alzheimer's disease and related dementias compared to the general population. Even after controlling for known risk factors, a separate analysis found that dementia occurred significantly more frequently in individuals with ASD alone, with an adjusted hazard ratio approaching 2.0. These are not marginal statistical differences; they suggest a biological connection running deep beneath the surface of both diagnoses.
The Genetic Overlap
At the molecular level, researchers have mapped a set of genes whose mutations appear in both conditions. Genes and proteins linked with both disorders include MECP2, ADNP, SCN2A, NLGN, SHANK, PTEN, RELN, and FMR1. These genes intersect at several key biological functions: regulating how neurons form and prune their connections, controlling excitatory and inhibitory balance in neural circuits, and governing chromatin structure, the physical packaging of DNA that determines which genes get switched on or off during brain development.
The ADNP gene, for instance, is among the most frequently mutated in autism; it is also emerging as a player in tau protein regulation, the hallmark of Alzheimer's neurodegeneration. PTEN mutations, well-documented in autism, are also implicated in the abnormal cell-growth signaling seen in Alzheimer's tissue. The overlap is not coincidental. Theories about the role of neuron development, processing, connectivity, and neurotransmitter levels have been discussed as common threads across both disorders.
A 2025 Frontiers in Neuroscience paper synthesizing this body of evidence identified convergent disruptions across two particularly consequential systems: the mTOR signaling network and the synaptic regulation machinery. Recent research points to significant genetic and biological overlaps suggesting shared pathways like the mTOR signaling system and synaptic regulation may increase risk for both conditions.
The mTOR Pathway: A Shared Molecular Highway
The mechanistic target of rapamycin, known as mTOR, is a nutrient-sensing protein that sits at the intersection of cell growth, autophagy, and synaptic maintenance. In autism, its disruption is well established: postmortem studies have revealed increased density of excitatory synapses in the brains of individuals with ASD, with a link to aberrant mTOR-dependent synaptic pruning. The brain, in effect, fails to trim its own circuitry during development, leaving synaptic connections overcrowded and poorly regulated.
In Alzheimer's disease, mTOR dysfunction operates at the opposite end of the lifespan but through a related mechanism. An overactive mTOR suppresses autophagy, the cellular process by which neurons clear out misfolded proteins including amyloid-beta and tau. When autophagy stalls, these toxic aggregates accumulate. Studies of Alzheimer's mouse models have shown that rapamycin, an mTOR inhibitor, can prevent or even reverse cognitive deficits, reduce levels of these toxic proteins, and improve neuroplasticity.
The significance of this shared pathway for drug development is difficult to overstate. A compound targeting mTOR dysregulation in autism might, with appropriate modification, serve as a therapeutic lever in early Alzheimer's progression. Conversely, insights gained from clinical trials of mTOR inhibitors in Alzheimer's patients could accelerate development of treatments for autistic adults at elevated dementia risk.
APOE and the Lymphatic System
A second convergence point involves the APOE gene, particularly the risk variant known as APOE ε4. Long established as the strongest genetic risk factor for late-onset Alzheimer's, APOE ε4 disrupts the brain's meningeal lymphatic system, impairing the drainage of amyloid-beta from brain tissue. The APOE ε4 allele disrupts meningeal lymphatic function and reduces amyloid beta clearance in Alzheimer's disease, while genetic factors in the same pathway have also been implicated in autism risk. This intersection suggests that impaired glymphatic clearance, a system that works primarily during sleep to flush metabolic waste from the brain, may represent a vulnerability shared across both diagnoses.
Rethinking Brain Development and Degeneration
The traditional clinical framing of autism as a disorder of "too much" connectivity and Alzheimer's as a disorder of "too little" has long made the two seem biologically incompatible. The emerging research complicates that binary. Both conditions involve disrupted synaptic maintenance, both implicate chronic neuroinflammatory processes, and both feature abnormalities in how the brain handles protein clearance and cellular cleanup.
A 2025 Swedish population-based family study found evidence of shared familial influences between autism and dementia across generations, suggesting that the elevated dementia risk in autistic individuals is not simply an artifact of co-occurring conditions or medication exposure, but reflects something heritable and biological at the root. This kind of multi-generational evidence points researchers toward upstream molecular targets rather than symptom-level interventions.
What This Means for Screening and Clinical Practice
The practical implications for clinicians are significant, if still developing. Given that autistic adults reach Alzheimer's-related dementia diagnoses in their late fifties on average, and given a prevalence rate eight times that of the general population, routine cognitive monitoring beginning in midlife could catch deterioration far earlier than current protocols allow. Clinicians who treat autistic adults for behavioral or mental health concerns may need to incorporate structured cognitive assessments as a standard part of care, something most current frameworks do not require.
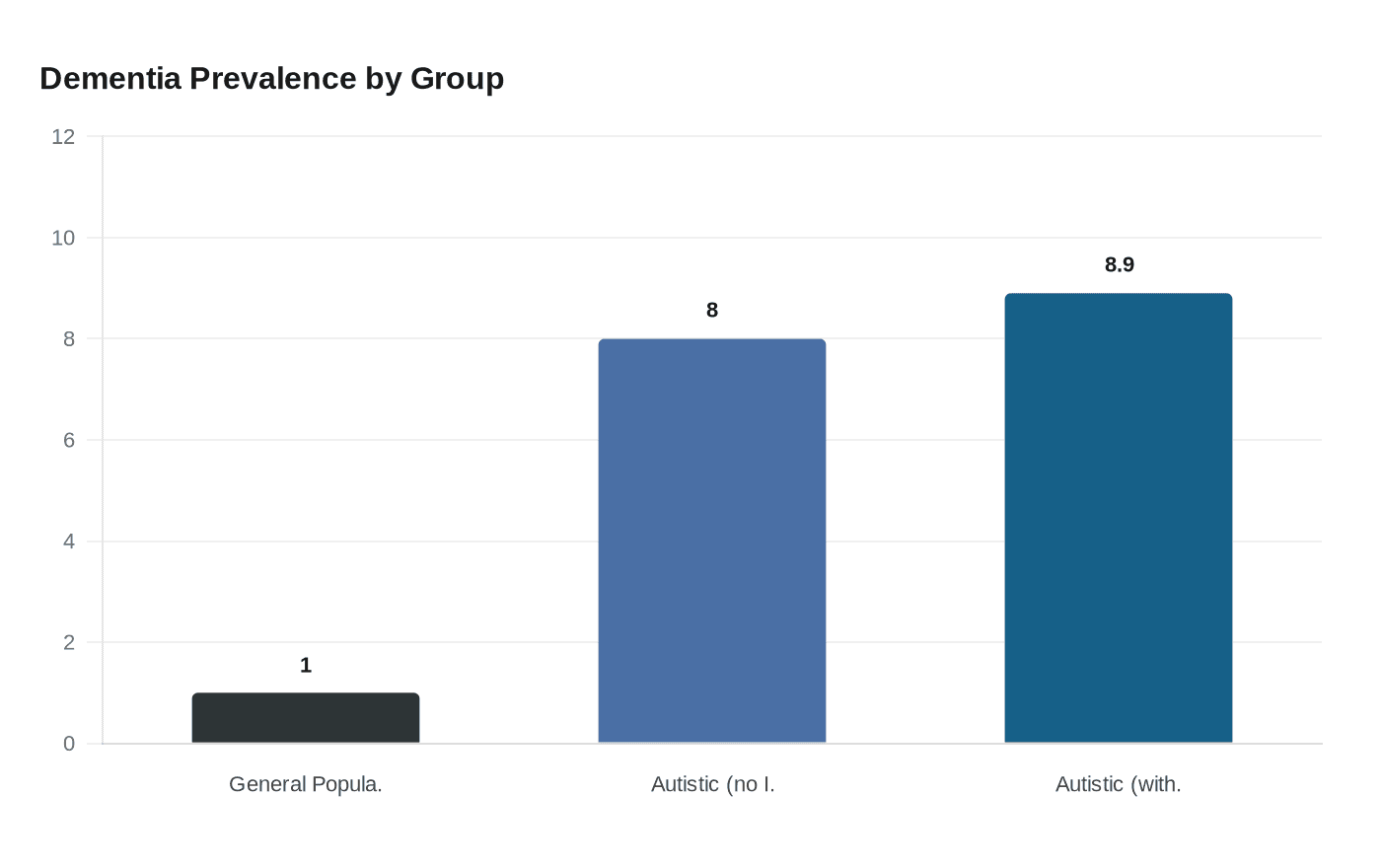
For families already navigating autism diagnoses, these findings carry weight that demands careful handling. The elevated risk is real and worth knowing about. It is not a certainty, and the research is still at an early, observational stage. The genetic and pathway overlaps identified so far explain correlation, not causation. No one should interpret a family member's autism diagnosis as a guaranteed trajectory toward Alzheimer's, and clinicians must guard against framing this research in ways that add unnecessary fear to families already managing complex caregiving demands.
The Drug Development Opportunity
Where the science does offer genuine hope is in the arena of therapeutic repurposing. A 2025 review in *Translational Psychiatry* examined how existing Alzheimer's disease medications perform in children and adolescents with autism and intellectual disability, finding early signals of benefit in neurocognitive outcomes. Meanwhile, the research pipeline for mTOR-targeting compounds is advancing on both sides of the diagnostic divide.
A 2025 paper in the *International Journal of Molecular Sciences* explicitly reconstructed gene networks linking ASD and Alzheimer's disease to identify opportunities for drug repurposing at the intersection of both conditions. The logic is straightforward: if the same molecular pathways drive pathology in both disorders, then compounds that correct those pathways need not be developed from scratch for each disease separately. A drug that succeeds in one context could move faster through trials in the other, potentially compressing development timelines that currently stretch over decades.
What Remains Unknown
The current evidence base is observational and mechanistic; no randomized controlled trial has yet tested a shared-pathway treatment across both diagnoses simultaneously. The gene networks identified, while compelling, represent associations rather than proven causal chains. Neuroinflammation, synaptic dysfunction, and protein clearance failures are each multi-factorial processes shaped by environment, lifestyle, and co-occurring health conditions as much as by genetics.
The field is also grappling with a fundamental measurement problem: cognitive decline in autistic adults is notoriously difficult to assess because standard dementia screening tools were not designed for people whose cognitive and communicative profiles differ significantly from neurotypical baselines. Better biomarkers, including blood-based assays for amyloid and tau that have transformed Alzheimer's detection in the general population, will need to be validated specifically in autistic cohorts before they can guide clinical decisions.
The science connecting autism and Alzheimer's is still assembling its pieces, but the convergence is no longer a peripheral hypothesis. It is reshaping how neuroscientists think about the entire arc of brain health, from the first synapses formed in infancy to the last ones lost in old age, and offering a framework in which treating one condition better may, at last, mean treating the other better too.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip