Stereotaxis Wins FDA Clearance for Synchrony, a 4K Digital Cockpit for Cardiac Cath Labs
Stereotaxis secured FDA 510(k) clearance for Synchrony, a 55-inch 4K cath-lab display that targets every interventional lab, not just robotic ones, with $3M in 2026 revenue in its sights.
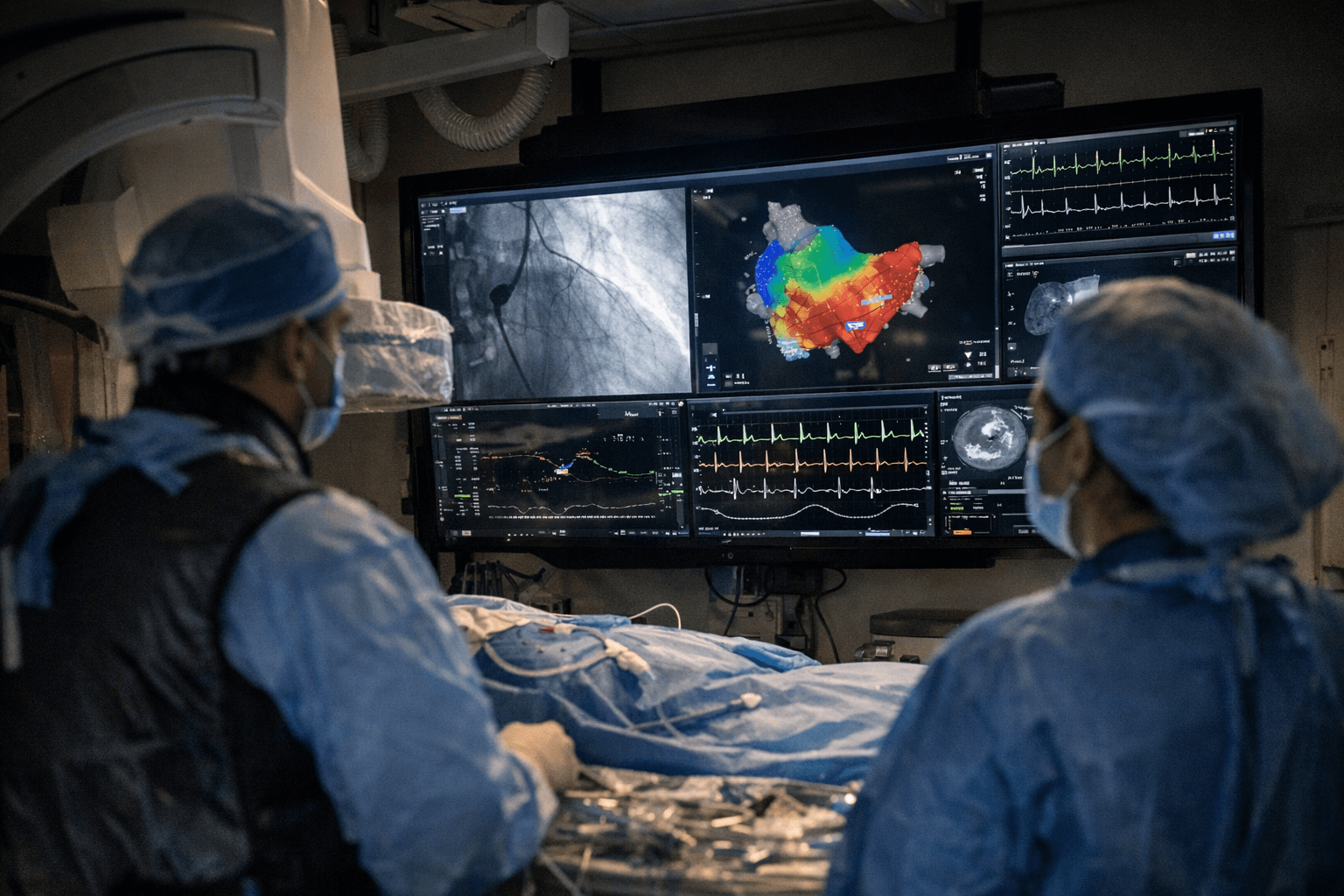
A catheterization lab today is a patchwork of screens: one monitor for fluoroscopy, another for mapping, a third for hemodynamic data, and separate displays for whatever device each vendor requires. Stereotaxis, the St. Louis-based surgical robotics company trading on NYSE as STXS, received FDA 510(k) clearance on April 6, 2026 for Synchrony, a system built on the premise that all of those feeds should collapse into a single, 55-inch, 4K ultra-high-definition surface.
That FDA pathway matters for context. A 510(k) clearance does not establish that a device produces new clinical benefits or improves outcomes over existing alternatives. It establishes substantial equivalence to a legally marketed predicate device, meaning Synchrony clears a safety and effectiveness bar, but peer-reviewed evidence that it shortens procedure times, reduces radiation exposure, or cuts error rates has yet to be published. That real-world proof will largely determine whether hospital procurement committees write the checks.
What Synchrony proposes to change in practice is the cognitive architecture of an interventional procedure. The display digitizes all video streams with less than one frame of latency, offers custom layouts across imaging, mapping, and device controls, and is built, according to Stereotaxis, with "obsolescence protection" so hospitals are not locked out as new technologies arrive. The goal is a decluttered control environment during high-risk cardiac and electrophysiology cases where clinician attention is the scarcest resource.
Bundled with the display is SynX, a cloud-based application that is both HIPAA- and GDPR-compliant and enables secure remote connectivity, collaboration, recording, and monitoring of cath lab activity. The remote layer has direct implications for training and proctoring models: an experienced electrophysiologist in one city could observe, advise on, or co-navigate a case from another institution without traveling. Dr. Mauricio Arruda, professor of medicine at Case Western Reserve University School of Medicine and director of electrophysiology at University Hospitals Harrington Heart & Vascular Institute, called out that potential directly, saying his team has "long recognized that seamless remote connectivity and collaboration for our EP labs could be very beneficial" and positioned University Hospitals as an early adopter of both Synchrony and SynX.
The financial framing is equally significant. Stereotaxis reiterated guidance of more than $3 million in Synchrony revenue for 2026, explicitly placed independent of its robotic navigation systems. That independence is the strategic signal: Stereotaxis has historically sold to a narrow universe of labs willing to invest in robotic catheter navigation, but Synchrony targets every interventional lab, a market several orders of magnitude larger. Shares moved higher in premarket trading following the announcement as investors processed that expanded addressable market.
Chairman and CEO David Fischel described the ambition plainly: "The opportunity is much broader than robotic labs as we believe all cath labs stand to benefit from improved workflow, connectivity, and collaboration." Fischel also framed Synchrony as the connective tissue for future autonomous capabilities, noting the platform "will be critical in supporting robotic efforts for remote long-distance procedures and automated catheter navigation."
The Synchrony clearance is the third regulatory milestone for Stereotaxis in roughly six months. The company obtained CE Mark in Europe in October 2025, the FDA cleared its MAGiC Magnetic Interventional Ablation Catheter in January 2026, and now Synchrony completes what the company describes as its vision for the robotic digital cockpit. Together, those approvals suggest a deliberate build toward a more integrated hardware-software-cloud stack, one where recurring revenue from SynX subscriptions and software updates could eventually supplement or outpace equipment sales.
For hospitals evaluating adoption, the unanswered questions are practical. Interoperability with entrenched vendor ecosystems, cybersecurity architecture around a cloud-connected cath lab app, training burden for staff accustomed to proprietary displays, and the absence of prospective outcome data will all shape purchasing timelines. Synchrony's commercial launch resolves the regulatory question. Whether it resolves the workflow and clinical evidence questions is the test that follows.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

