Study Finds Cushing’s Pill May Extend Lives in Ovarian Cancer
A Cushing’s pill cut death risk by 35% in a 381-patient ovarian cancer trial, and the FDA has already approved it with chemotherapy.

For women with platinum-resistant ovarian cancer, a pill already used in Cushing’s syndrome is now showing the kind of survival gain that can matter most when standard chemotherapy has run out of room. In a phase 3 trial of 381 patients, relacorilant was linked to a 35% lower risk of death than usual care, a finding that comes in a disease where many patients live only about a year to 18 months after diagnosis.
The randomized controlled ROSELLA study tested relacorilant with nab-paclitaxel and found that the combination prolonged both progression-free survival and overall survival. The Lancet said the benefit came without biomarker selection, meaning the treatment did not depend on finding a molecular subgroup before starting therapy. Sky News reported that the median overall survival improved by 4.1 months, a difference that is modest in absolute terms but meaningful in a cancer that has already stopped responding to platinum-based chemotherapy within six months of treatment.
That survival signal has already moved beyond the trial setting. On March 25, 2026, the U.S. Food and Drug Administration approved relacorilant, sold as Lifyorli, in combination with nab-paclitaxel for adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer after one to three prior systemic regimens, at least one of which included bevacizumab. The agency describes relacorilant as a glucocorticoid receptor antagonist. Corcept Therapeutics said the ROSELLA data were presented at the Society of Gynecologic Oncology’s 2026 annual meeting, published simultaneously in The Lancet, and led to the combination being added to the National Comprehensive Cancer Network guidelines as a preferred regimen.
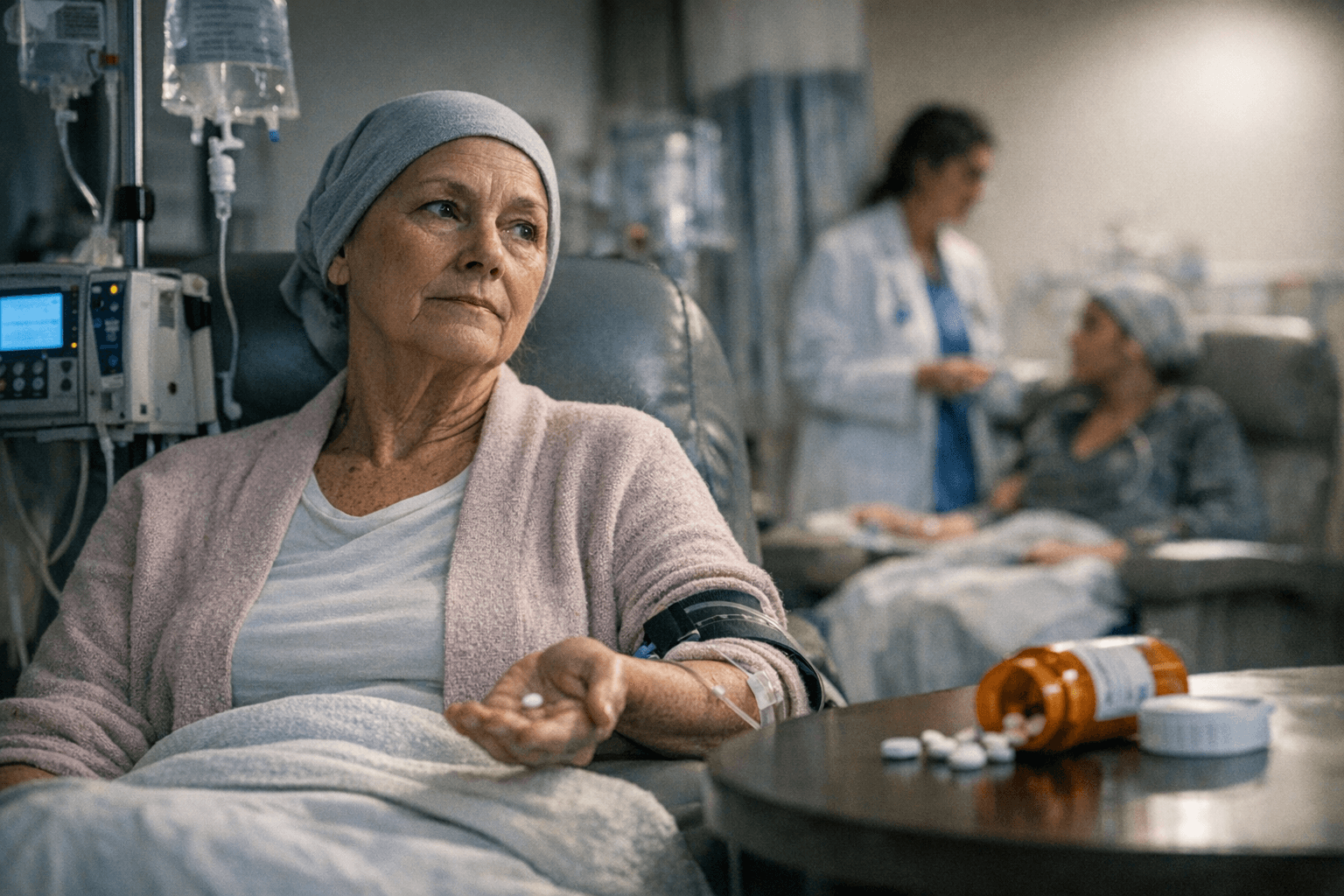
The result stands out because relacorilant is not a brand-new cancer molecule built from scratch. It is a repurposed drug with an existing medical history, which can shorten the path from study to bedside when the evidence is strong enough. That matters in ovarian cancer, where patients often enter later lines of therapy with limited options and little time to wait for an entirely new drug development cycle.
The same Lancet issue also highlighted another late-stage advance for the same hard-to-treat population. A phase 3 study found that pembrolizumab plus weekly paclitaxel, with or without bevacizumab, significantly improved progression-free survival and overall survival in platinum-resistant recurrent ovarian cancer. Roswell Park Comprehensive Cancer Center in Buffalo, New York, said those results helped pave the way for U.S. and European approvals, underscoring a broader shift toward incremental but real progress in a disease that has long offered too few choices.
Know something we missed? Have a correction or additional information?
Submit a Tip
