Trump Eyes 100% Tariff on Imported Brand-Name Drugs to Cut Prices
The Trump administration prepared a 100% tariff on patented drug imports, giving big pharma 120 days to sign pricing deals or commit to U.S. production.

The Trump administration moved to impose a 100% tariff on branded, patented pharmaceuticals imported into the United States, giving the world's largest drugmakers a stark choice: negotiate price cuts with the federal government, pledge to build production facilities on American soil, or face a duty that would effectively double the landed cost of their medicines in this market.
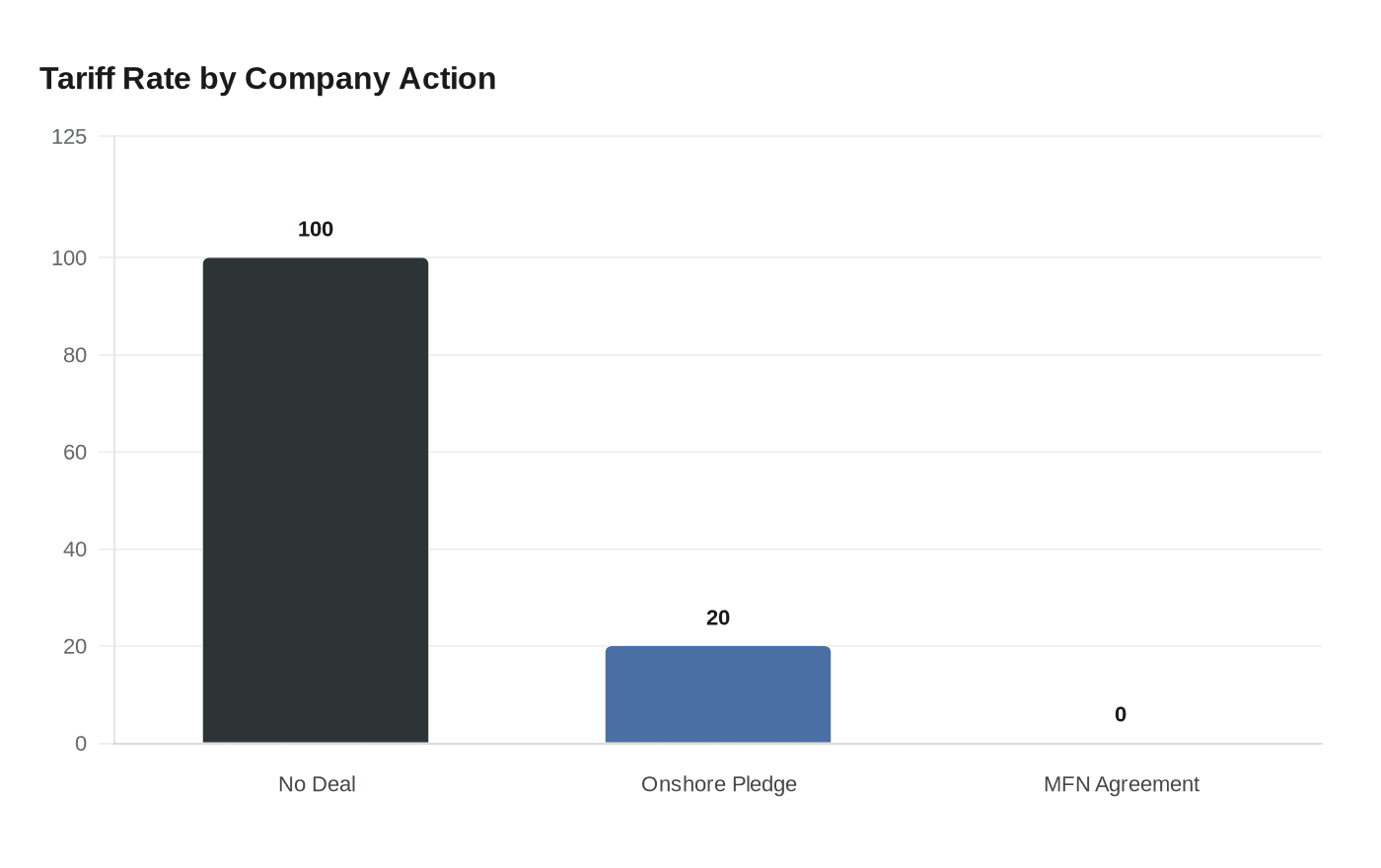
Under the terms of the proposed order, patented drugs not manufactured domestically and not covered by a government pricing agreement would face the full 100% levy. Companies that commit to onshoring production would qualify for a reduced 20% tariff. Those that sign most-favored-nation pricing agreements with the Department of Health and Human Services could be exempt entirely. The administration set tiered deadlines: larger pharmaceutical companies would have 120 days to outline plans to avoid the tariff, while smaller firms would have 180 days to do the same.
The measure would not apply to most generic drugs, which already account for the majority of U.S. prescriptions. Instead, the policy zeroes in on brand-name manufacturers, the companies that set list prices for patented medicines and routinely cite high research-and-development costs to justify charging American patients far more than their counterparts in other high-income countries pay for the same therapies.
The administration was not starting from zero on negotiations. About 17 pharmaceutical companies already had drug-pricing arrangements with the White House, with 13 of those deals finalized. The 100% tariff framework would sharpen pressure on the holdouts, while industry groups were exploring individual arrangements and some companies were seeking carve-outs or phased timelines to soften the impact.
The proposal carries substantial legal and diplomatic weight. Critics warned that a tariff of this magnitude could disrupt global pharmaceutical supply chains, trigger retaliatory measures from major trading partners, and raise costs in countries without their own negotiated deals. It also raised compliance questions under World Trade Organization rules. Supporters, however, argued that without a lever of this size, manufacturers faced little structural incentive to lower prices that have remained elevated in the U.S. relative to the rest of the developed world.
If finalized, the order would represent the most aggressive move yet in the administration's effort to lower U.S. drug costs and re-shore production of active pharmaceutical ingredients and final dosage forms. Pharmaceutical companies will need to quickly assess whether to sign pricing deals with HHS, accelerate investment in domestic manufacturing capacity, or mount a legal challenge before the deadlines arrive.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip