Trump orders faster psychedelic research, access for mental illness treatment
Trump moved to speed psychedelic research and patient access, but not legalization, betting $50 million and federal review can turn ibogaine into a mental-health treatment.

Donald Trump used an executive order to push psychedelic medicine closer to mainstream federal review, but stopped short of legalizing anything. The April 18 order, titled Accelerating Medical Treatments for Serious Mental Illness, told the Food and Drug Administration to prioritize psychedelic drugs that have already won Breakthrough Therapy designation and instructed the FDA and Drug Enforcement Administration to build a pathway for eligible patients to receive investigational drugs, including ibogaine compounds, under the administration’s Right to Try framework.
That is the key distinction in the policy shift: the White House is opening doors to research access and faster review, not removing federal drug controls. Ibogaine remains a Schedule I substance under federal law, a category reserved for drugs the government says have no currently accepted medical use and a high potential for abuse. The administration, however, said there is now enough scientific evidence to justify deeper study, especially for veterans and people with hard-to-treat conditions such as post-traumatic stress disorder, depression, opioid addiction and traumatic brain injury.
The White House said more than 14 million American adults have a serious mental illness and pointed to veteran suicide rates that have exceeded 6,000 per year for more than 20 years. It also said it would dedicate $50 million to federal research into ibogaine, signaling that the order is meant to move beyond symbolism and into laboratory and clinical work. FDA Commissioner Marty Makary said decisions on the drugs could come as soon as this summer, setting an unusually fast timetable for a field that has been stalled for decades by stigma, regulation and limited large-scale data.
Trump unveiled the order in the Oval Office with Joe Rogan, Makary, and veteran advocates including former Navy SEAL Marcus Luttrell and Rep. Morgan Luttrell, a lineup that underscored the political strategy behind the announcement. The move followed earlier signs that the administration was warming to psychedelic medicine, including public support from Health and Human Services Secretary Robert F. Kennedy Jr. It also fits into a broader drug-policy push that began in December 2025, when Trump signed an executive order directing the attorney general to expedite rescheduling marijuana to Schedule III.
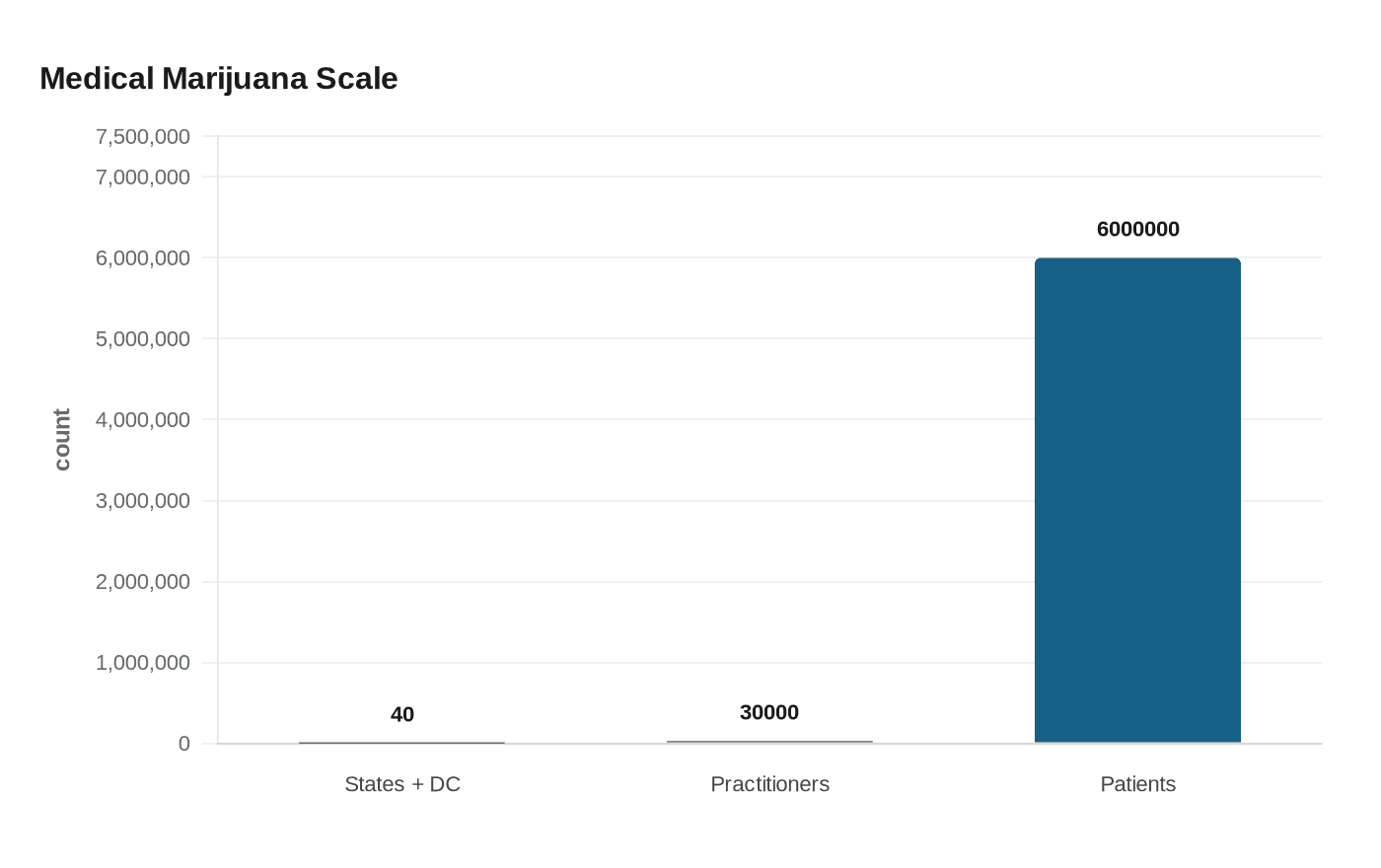
That marijuana order cited 40 states plus the District of Columbia with regulated medical marijuana programs and said 30,000 licensed health care practitioners are authorized to recommend medical marijuana for more than six million registered patients. Together, the two orders suggest a White House willing to use federal agencies, scheduling reviews and research channels to loosen the grip of drug prohibition without calling it legalization. For patients, though, the practical effect still depends on how quickly the FDA, the DEA and the medical evidence can move from announcement to access.
Know something we missed? Have a correction or additional information?
Submit a Tip

