U.S. fast-tracks Medicare coverage for breakthrough medical devices
Medicare coverage for breakthrough devices could now arrive in about two months instead of a year or more. Regulators say the RAPID pathway may speed access for roughly 40 devices.

Medicare coverage for certain breakthrough devices could now arrive in about two months instead of a year or more, a shift regulators say will bring seniors new implants, monitors and diagnostic tools far sooner after FDA authorization.
The Centers for Medicare & Medicaid Services and the Food and Drug Administration announced the RAPID pathway on April 23, 2026, a program built to align FDA approval with Medicare reimbursement decisions for selected Breakthrough Devices that address unmet medical needs among Medicare beneficiaries. RAPID stands for Regulatory Alignment for Predictable and Immediate Device coverage. CMS said the pathway will cover certain Class II devices participating in the FDA’s Total Product Life Cycle Advisory Program, or TAP, and Class III devices whether or not they participate in TAP.
Under the new process, CMS would issue a proposed national coverage determination on the same day the FDA grants market authorization. That would be followed by a comment period, while a Federal Register notice describing the pathway will also be published for 60 days of public comment. Regulators said the aim is to reduce the long gap between approval and reimbursement, a delay device makers say can stall hospital adoption, discourage investment and leave patients waiting after a product has already cleared a major evidence and innovation threshold.

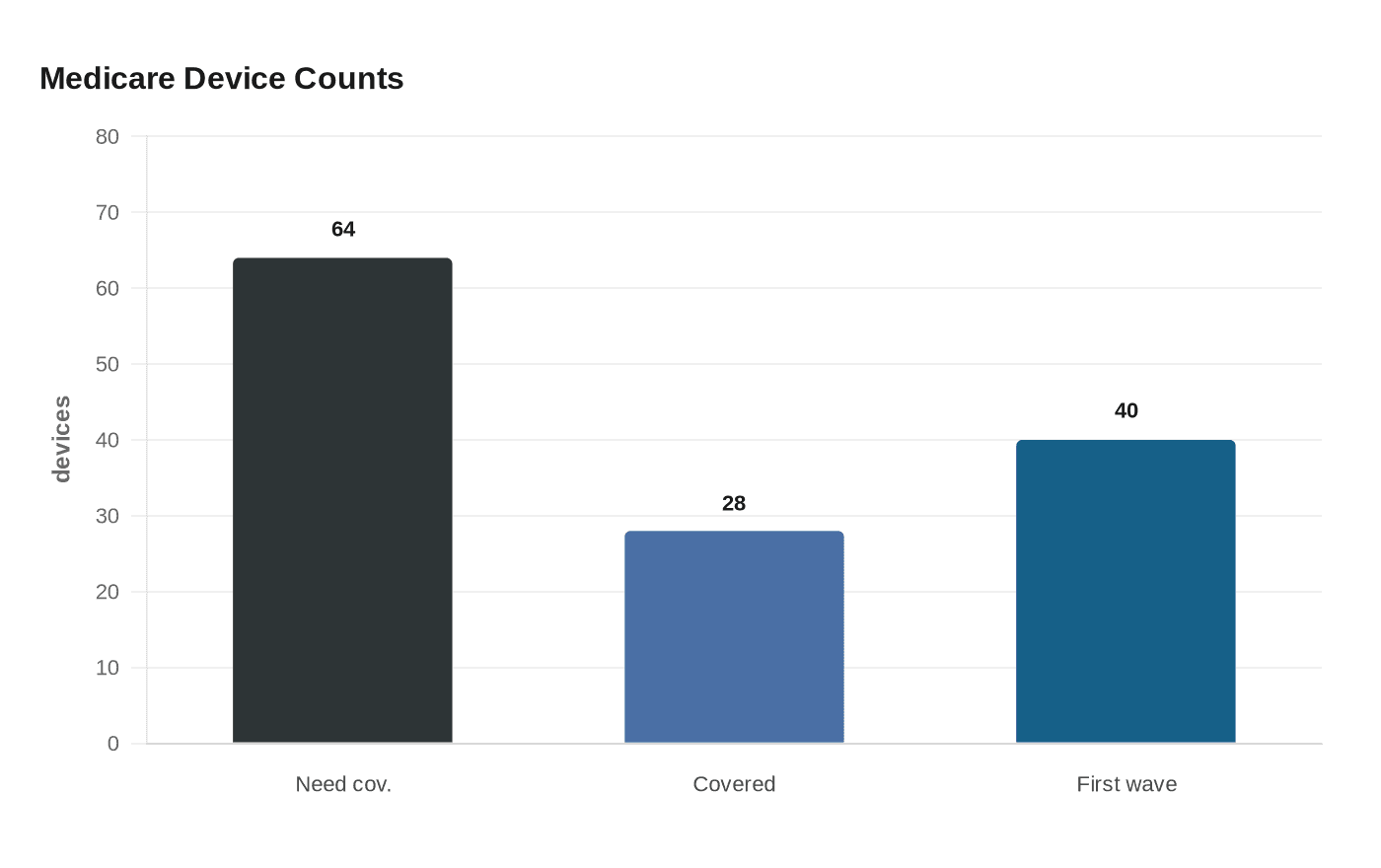
The timing matters because the Medicare lag has been measured in years, not months. A 2023 JAMA Health Forum study found that among 64 devices and diagnostics requiring new Medicare coverage, only 28, or 44%, had at least nominal coverage within a median of 5.7 years. Stanford researchers found the time from FDA authorization to a Medicare coverage milestone ranged from less than 91 days to about seven years. FDA’s Breakthrough Devices program began in 2016, and CMS issued the Transitional Coverage for Emerging Technologies procedural notice in 2024, but the new RAPID pathway is meant to synchronize the two agencies more tightly.
Dr. Mehmet Oz and FDA Commissioner Marty Makary both backed the plan on April 23, 2026, saying the agencies work best when they move together earlier in the process. The first wave is expected to include about 40 devices. The policy also lands as large manufacturers push for a clearer route to coverage, with Johnson & Johnson generating $33.8 billion in medtech sales in 2025 and Medtronic posting $33.5 billion in revenue.
The question now is whether faster Medicare decisions will shorten bureaucratic delay without weakening evidence standards, or open the door to expensive devices that win speed before they prove enough value.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

