U.S. panel considers delaying newborn hepatitis B vaccine, experts warn
A federal vaccine advisory committee led by Dr. Kirk Milhoan, appointed by Health Secretary Robert F. Kennedy Jr., said it would vote this week on whether to delay the hepatitis B vaccine for newborns, a move that could affect routine immunization schedules across the country. Public health specialists and manufacturers cautioned that shifting the policy could reverse decades of progress, aggravate supply logistics, and deepen health inequities for communities already at higher risk.
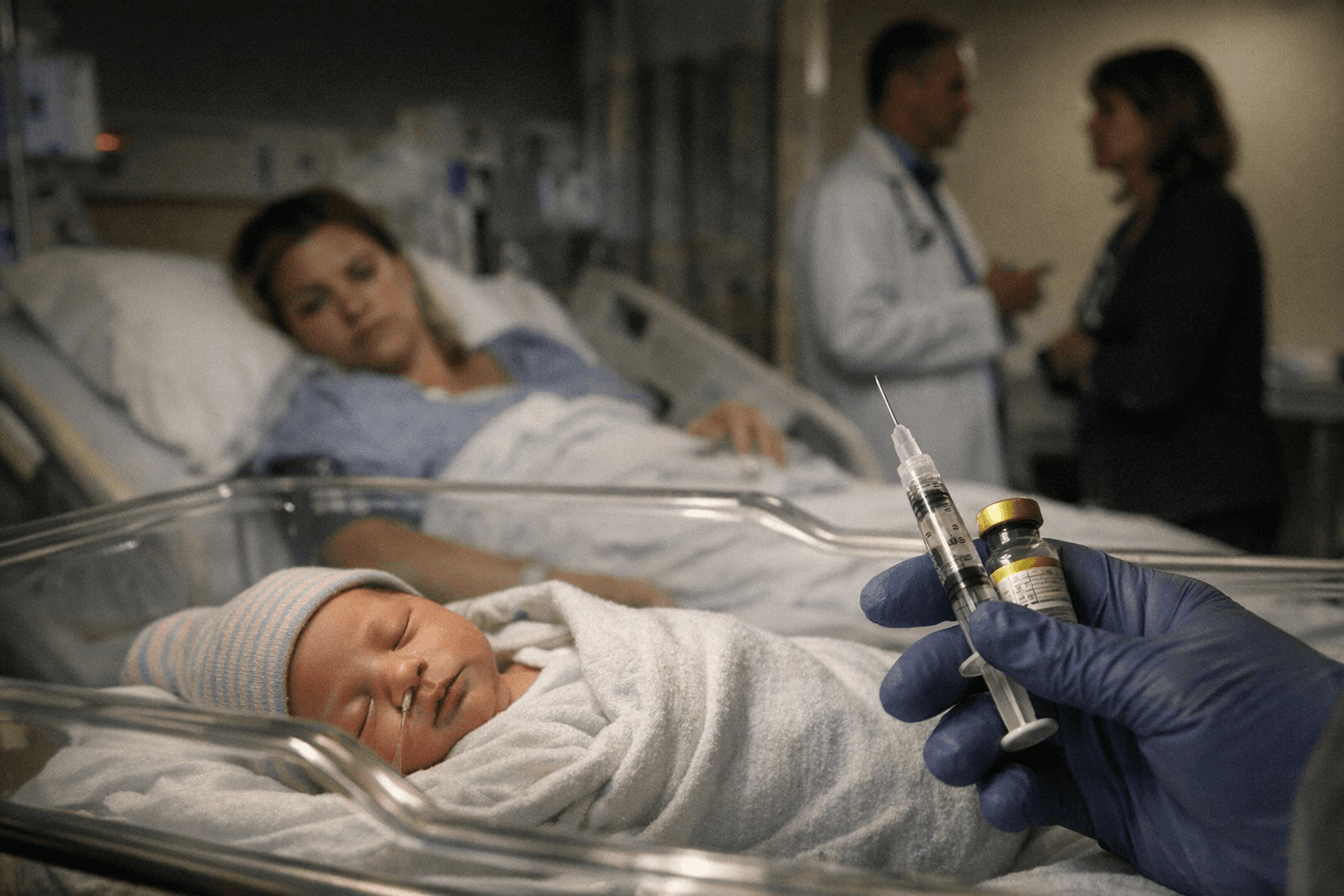
The federal vaccine advisory committee chaired by Dr. Kirk Milhoan, an appointee of Health Secretary Robert F. Kennedy Jr., notified colleagues on December 2 that it would vote this week on whether to delay administration of the hepatitis B vaccine for newborns. The panel has not settled on how long any delay might last, and the proposal is part of a broader review of immunization timing under Secretary Kennedy, who has previously questioned routine vaccine schedules.
The announcement came the same day a systematic review of more than 400 studies concluded that the long standing U.S. policy of giving the hepatitis B vaccine to newborns reduced infections in children by more than 95 percent. The finding reinforces public health arguments that immediate newborn vaccination is a cornerstone of preventing perinatal transmission of hepatitis B and of protecting infants who face a high risk of developing chronic infection if exposed at birth.
Vaccine manufacturers warned regulators that changing the newborn recommendation could create supply disruptions. Many vaccines are produced and distributed according to the established childhood immunization timetable, and manufacturers say alterations to timing would require adjustments to production planning and packaging, with potential ripple effects for clinics and public programs that serve children.
Public health officials and pediatric experts expressed concern that altering the recommendation could open a path to resurgences of a preventable illness. Hepatitis B acquired in infancy often leads to long term liver disease and contributes to disparities in health outcomes among communities with limited access to prenatal screening and pediatric care. Delaying the initial dose, critics say, could disproportionately expose infants born to parents who face barriers to early follow up care, including low income families, recent immigrants, and rural populations where continuity of pediatric services is less reliable.
The committee vote poses immediate questions about how federal agencies balance emerging scientific reviews, logistical realities of vaccine supply, and the social consequences of policy change. Advocates for maintaining current practice point to the substantial reduction in childhood infections recorded since the policy was widely adopted, and to the public health value of ensuring that every newborn receives at least an initial dose before discharge from the hospital.
Those urging flexibility emphasize parental choice and calls for more research into optimal timing, but they also face practical constraints if a delayed schedule were adopted. Combination vaccines and coordinated pediatric appointments are designed around existing schedules, meaning retooling could require time and investment that safety nets do not always provide to the communities that need them most.
As the advisory committee prepares to vote, the decision will extend beyond clinical nuance into questions of health equity, supply chain resilience, and trust in public health guidance. Federal officials will have to weigh the review findings and manufacturer cautions against the potential for widening inequities in disease prevention among the country’s most vulnerable newborns.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

