VDyne Wins FDA Clearance to Launch Pivotal Tricuspid Valve Replacement Trial
VDyne secured FDA clearance to launch the TRIVITA tricuspid valve replacement trial, targeting 1.5 million U.S. patients too frail for open-heart surgery.

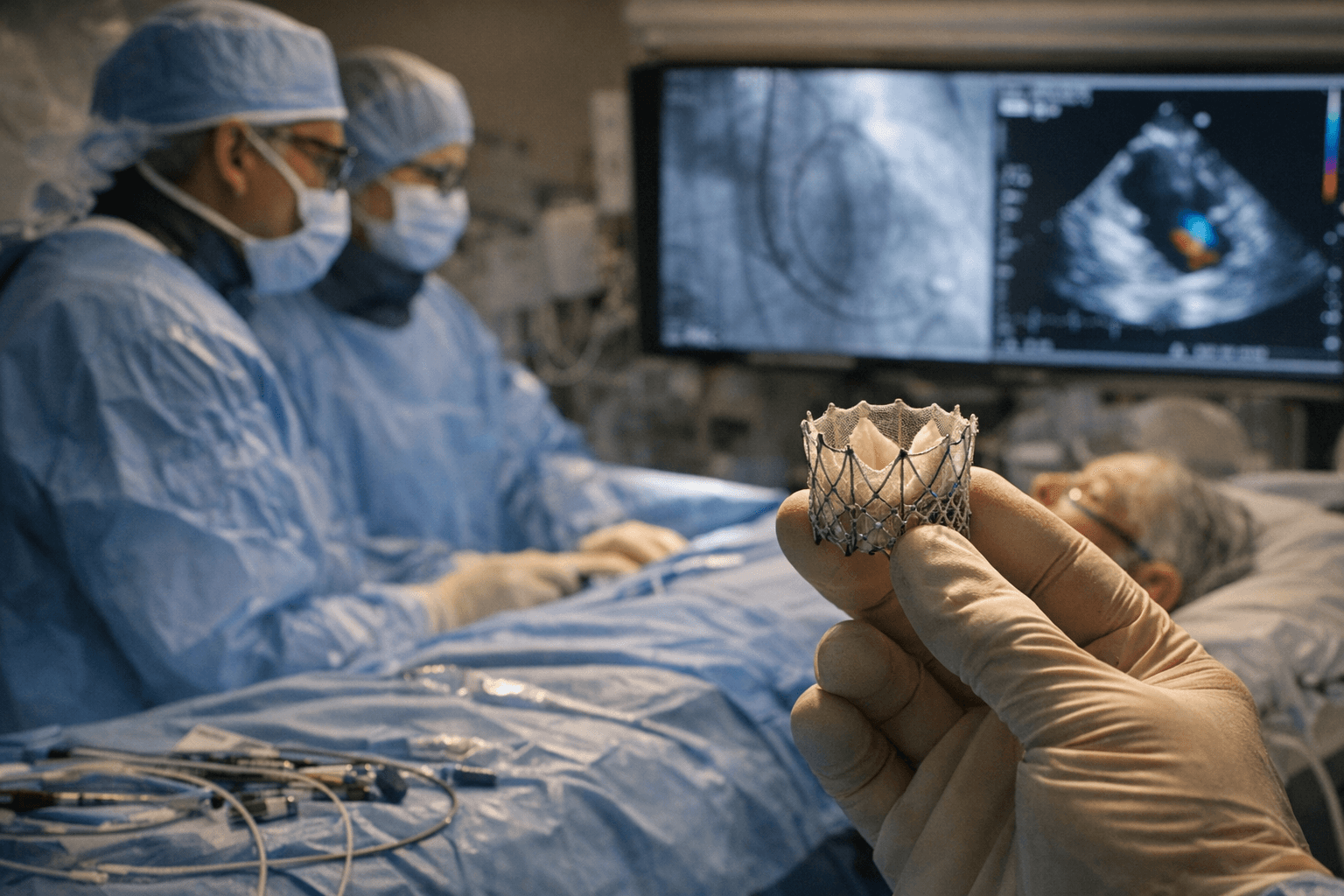
VDyne Medical secured FDA authorization to begin a pivotal U.S. clinical trial of its transcatheter tricuspid valve replacement system, placing the medical device startup in direct competition with Edwards Lifesciences' Evoque, currently the only FDA-authorized device of its kind in the space.
The FDA granted VDyne an investigational device exemption, the regulatory pathway required before launching a pivotal study in the United States. The trial, named TRIVITA, will enroll patients with symptomatic severe tricuspid regurgitation at leading U.S. clinical centers. The condition occurs when the tricuspid valve fails to close properly, allowing blood to leak backward into the right atrium and placing progressive strain on the heart. Roughly 1.5 million Americans live with the disease, and a substantial share of them cannot safely undergo open-heart surgery because conventional repair and replacement carry elevated mortality risk in this typically frail population.
VDyne said the TRIVITA study is designed to generate the safety and efficacy data required for a future full regulatory approval and broader clinical adoption. The company framed the IDE as a concrete step toward filling an access gap in a disease it described as historically undertreated.
Edwards Lifesciences' Evoque became the first FDA-authorized transcatheter tricuspid valve replacement in 2024, giving it a head start in a market defined by steep technical and training barriers. Medtronic's Intrepid device remained in early feasibility testing as of the announcement, while Abbott's TriClip, a repair rather than a replacement technology, is already on the market. VDyne's authorization positions the company as a direct competitor in the replacement segment, where approved options remain scarce relative to patient need.
To support both trial execution and eventual commercialization, VDyne recently appointed Mike Buck, previously chief executive of Verge Medical, as its chairman and CEO.
If TRIVITA produces positive outcomes, the data could reshape treatment algorithms at cardiology and cardiac surgery centers, prompt competitive responses from Medtronic and other companies in feasibility stages, and alter referral patterns for patients who currently lack minimally invasive replacement options. FDA approval in structural heart disease tends to confer durable commercial advantages, given the specialized training catheter-based systems require.
Tricuspid regurgitation has been an under-recognized driver of heart failure hospitalizations for years. VDyne's trial authorization moves the field one step closer to offering a second transcatheter replacement option in a space where the gap between patient need and available technology has long been the defining clinical constraint.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

