Vertex profit beats estimates as Alyftrek sales surge nearly eight-fold
Alyftrek sales jumped to $424.4 million, helping Vertex beat profit estimates as it pushed deeper into a broader business beyond cystic fibrosis.

Vertex Pharmaceuticals beat first-quarter profit expectations as a fast-ramping launch of Alyftrek added fresh momentum to a company still anchored by cystic fibrosis. The Boston-based drugmaker said Alyftrek generated $424.4 million in quarterly sales, up from $53.9 million a year earlier, a surge that underscores both the demand for better CF treatment and the commercial power Vertex still holds in the disease.
The gains were broad enough to lift total revenue to $2.99 billion, up 8% from a year earlier. U.S. revenue rose 7% to $1.78 billion, while revenue outside the United States climbed 9% to $1.21 billion. Vertex said Casgevy and Journavx accounted for more than 25% of the quarter’s growth, a sign that the company is trying to reduce its dependence on a single franchise even as cystic fibrosis remains the core of the business.

That transition is happening in a market shaped by medical progress and access limits. Cystic fibrosis is a rare, progressive genetic disorder caused by the absence of a protein that regulates salt and water transport in cells, leading to serious respiratory and digestive complications. Vertex said an April 1 label expansion extended Alyftrek and Trikafta to about 95% of people with CF in the United States, adding roughly 800 newly eligible patients for a CFTR modulator. The company said the expansion was supported by clinical or in vitro data from 564 variants for Alyftrek and 521 variants for Trikafta.
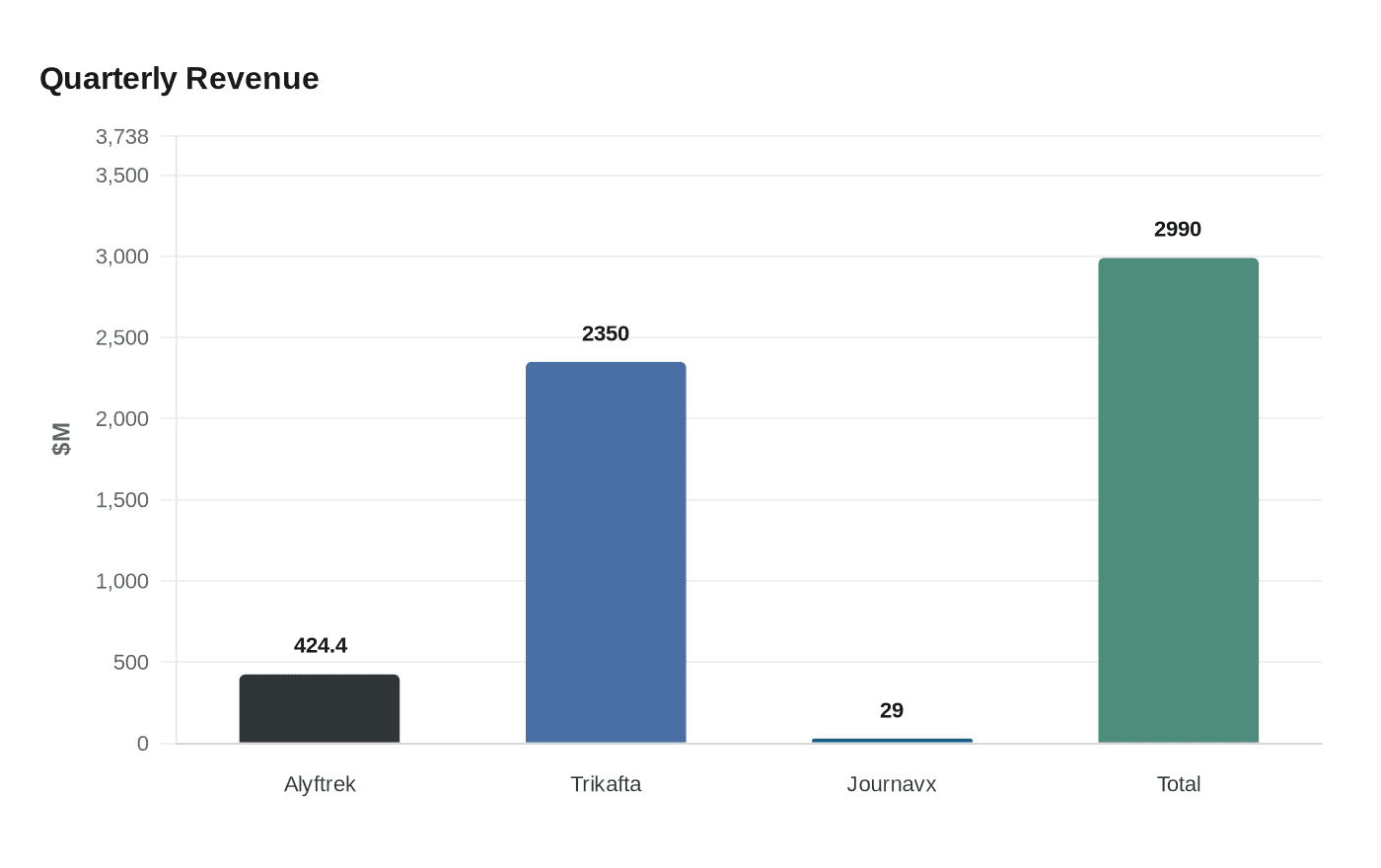
Even so, the company’s older CF therapy Trikafta still brought in $2.35 billion in quarterly sales, but that figure missed analyst expectations. For a dominant drug maker, that miss matters: it suggests payer pressure and market saturation can bite even when a franchise remains enormous. Vertex’s grip on CF gives it pricing power, but it also raises the stakes for insurers and health systems weighing the cost of lifelong treatment against the clinical benefit of broader genetic eligibility.
Vertex is trying to show that its future can stretch beyond CF. Journavx, a non-opioid pain treatment launched in the U.S. at the start of 2025, generated $29 million in first-quarter revenue and more than 350,000 prescriptions were filled. The Centers for Medicare and Medicaid Services approved its inclusion in the NOPAIN Act separate payment list, with a retroactive payment date of January 23, 2026, which should help speed hospital adoption.
Reshma Kewalramani said the company was "off to a strong start in 2026" and that "CASGEVY and JOURNAVX delivered more than 25 percent of our growth this quarter." Vertex reiterated full-year 2026 revenue guidance of $12.95 billion to $13.1 billion and said it aims to top $500 million in 2026 revenue from products outside CF, up from $175.6 million in 2025. It also pointed to progress on povetacicept in IgA nephropathy and new studies in primary membranous nephropathy and generalized myasthenia gravis, further evidence that Vertex is trying to build a second act while its CF engine still does the heavy lifting.
Know something we missed? Have a correction or additional information?
Submit a Tip
