Gene therapy rewires motor cortex, restores walking after spinal cord injury
A designer cytokine made motor-cortex neurons act like local protein factories, and mice with spinal contusions regained coordinated walking.
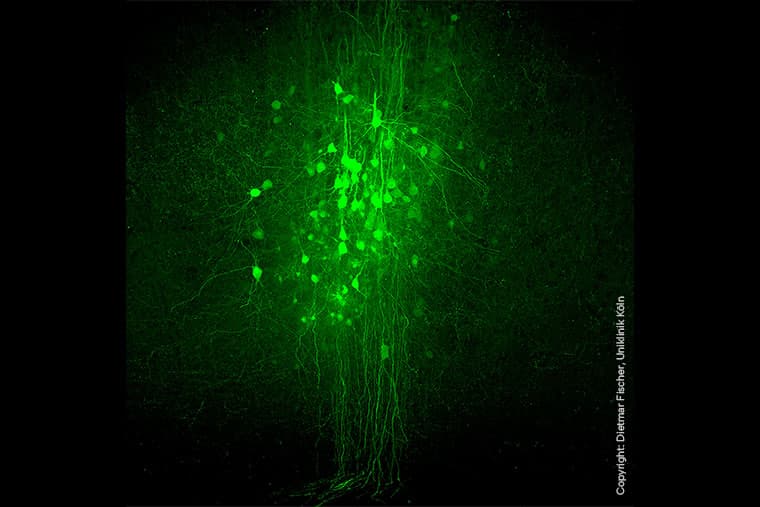
A single gene therapy turned the motor cortex into a protein-making relay, and that shift was enough to help mice with spinal cord contusions walk again. In the new work from Dietmar Fischer’s group at the Institute of Pharmacology II at University Hospital Cologne and Ruhr-Universität Bochum, nerve cells in the motor cortex were instructed to produce hyper-interleukin-6, a designer cytokine that does not occur naturally and must be built through genetic engineering.
The treatment used an adeno-associated virus vector injected into the motor cortex, or sensorimotor cortex, where it converted those neurons into a local source of hIL-6. From there, the signal was passed transneuronally to deeper motor regions, reshaping descending circuits rather than simply trying to regrow severed axons. The paper, titled Transneuronal cytokine delivery promotes functional recovery across spinal cord contusion severities via descending circuit plasticity, was already online ahead of its June 15, 2026 publication in Neurobiology of Disease.
The results reached across the injury spectrum that matters most in clinic. In mouse models of mild, moderate and severe spinal cord contusion, the treated animals showed significantly improved walking ability, including restored coordinated gait patterns. That detail matters because most human spinal cord injuries are contusions, where some fibers are damaged but others remain intact. The strategy is therefore less about rebuilding an entire broken highway and more about rerouting traffic through surviving roads.
Mechanistically, the study pointed to sprouting and network reorganization. The treatment did not reduce lesion size or overall nerve-cell loss, which argues that the benefit came from new collateral branches forming in intact descending pathways. Serotonergic neurons in the brainstem proved central to the recovery: when those cells were removed, the functional gains largely disappeared.

The work builds on a related 2021 study from the same group, published in Nature Communications, in which a single injection of AAV-hyper-interleukin-6 into the sensorimotor cortex produced corticospinal and raphe tract regeneration and functional recovery after severe spinal cord injury in mice. In that earlier experiment, previously paralyzed mice began to walk again about two to three weeks after treatment. The new study strengthens the case that the cortex can be used as an accessible launch point to drive hard-to-reach motor circuits.
The mouse data are striking, but the human test will be harder. Any therapy built on viral gene delivery has to clear safety questions around dose, targeting and long-term expression, and the rewired circuits will need to hold up over time. Chronic spinal cord injury adds another challenge: scarred tissue, older lesions and years of network remodeling may narrow the window for bypass logic. Even so, the new work suggests that for injuries where some descending fibers survive, the future may lie as much in protein engineering and circuit rewiring as in regeneration alone.
Know something we missed? Have a correction or additional information?
Submit a Tip

