HMGB1 protein spreads aging through blood, blocking it reverses tissue decline
A redox-sensitive HMGB1 signal moved aging traits through blood in mice, and blocking it restored muscle repair and performance.
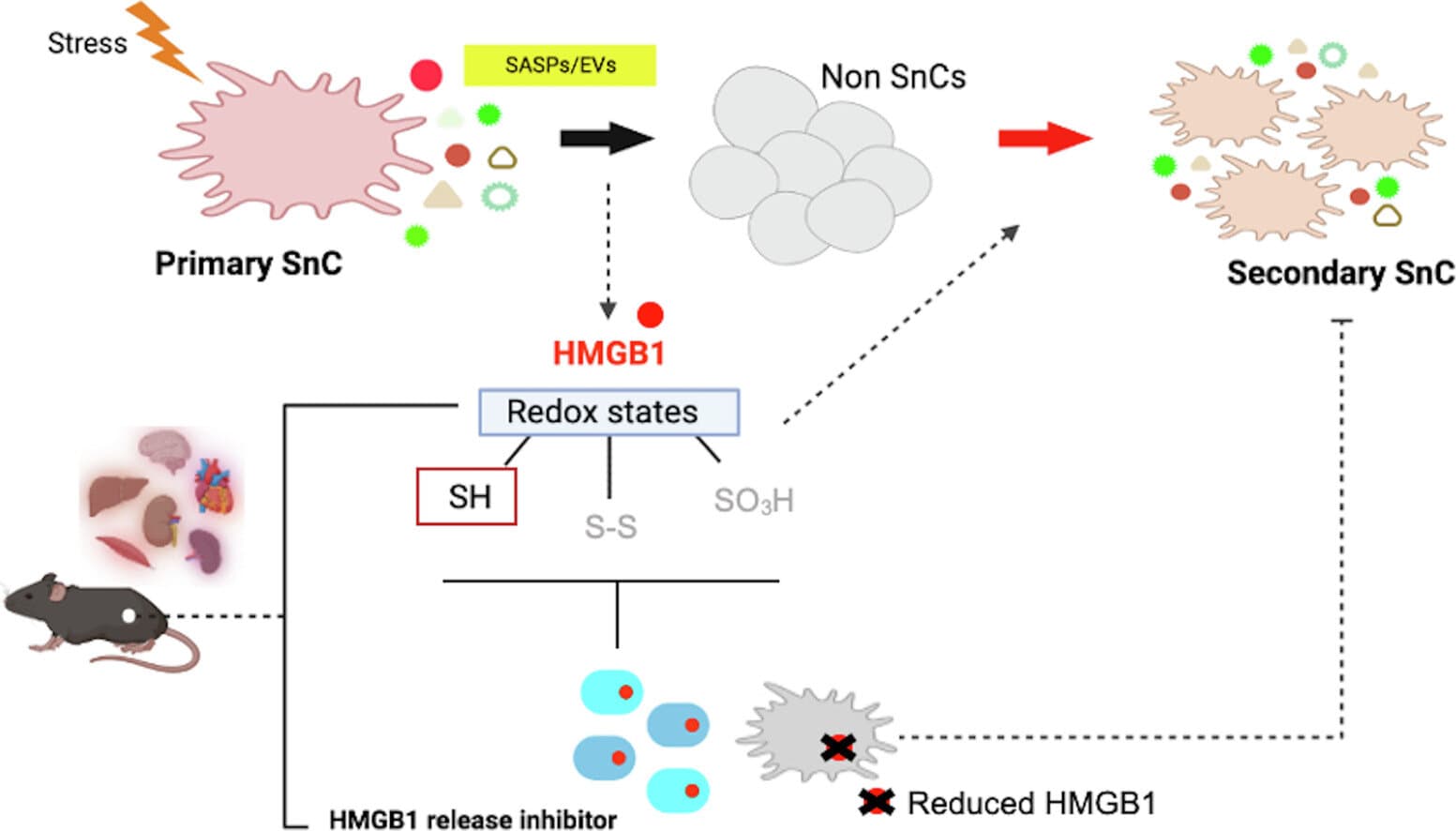
The key shift here is not another vague anti-aging claim but a blood-borne mechanism. Ok Hee Jeon’s group at Korea University College of Medicine reported that reduced HMGB1, or ReHMGB1, can circulate through the bloodstream and push remote tissues into a senescence-like state, giving the aging process a mobile signal instead of a purely local one.
The study, published in 2025 in Metabolism: Clinical and Experimental, volume 168, tested human fibroblasts, renal epithelial cells and skeletal muscle cells, then moved into mice. ReHMGB1, but not oxidized HMGB1, triggered senescence-like changes in the human cells and raised classic aging markers p21 and p16 in animals. In the mice, the protein also increased senescence-associated secretory phenotype, or SASP, factors and impaired muscle function, fitting the broader model in which senescent cells spread dysfunction by releasing inflammatory signals that weaken regeneration.
That redox split matters. Oxidized HMGB1 did not robustly produce the same effect, which makes the molecule less like a generic age marker and more like a switchable messenger. The paper, titled “Propagation of senescent phenotypes by extracellular HMGB1 is dependent on its redox state,” lists collaborators including Michael J. Conboy, Irina M. Conboy and Christopher D. Wiley, and the Korea University release says the work also drew on aging expertise from the University of California, Berkeley, and Tufts University.
The most practical result came in a middle-aged mouse muscle injury model. When researchers used anti-HMGB1 antibodies, senescence markers fell, muscle regeneration improved and physical performance recovered. That is the kind of preclinical result that gets attention because it points past theory and into tissue repair, but it is still a mouse result, not a therapy ready for clinics in Seoul, Berkeley or Medford. Human aging is messier, doses will matter, and blocking HMGB1 could carry risks because the protein also has normal roles in inflammation and repair.
Still, the translational angle is hard to ignore. Jeon’s team said this was the first evidence that a circulating HMGB1 isoform can induce senescence in remote tissues, and the finding opens a new way to think about disorders tied to aging biology, from muscle loss and frailty to conditions such as arthritis and Alzheimer’s disease. The real test now is whether dialing down HMGB1 in people can slow tissue decline without blunting the body’s own cleanup and healing systems.
Know something we missed? Have a correction or additional information?
Submit a Tip

