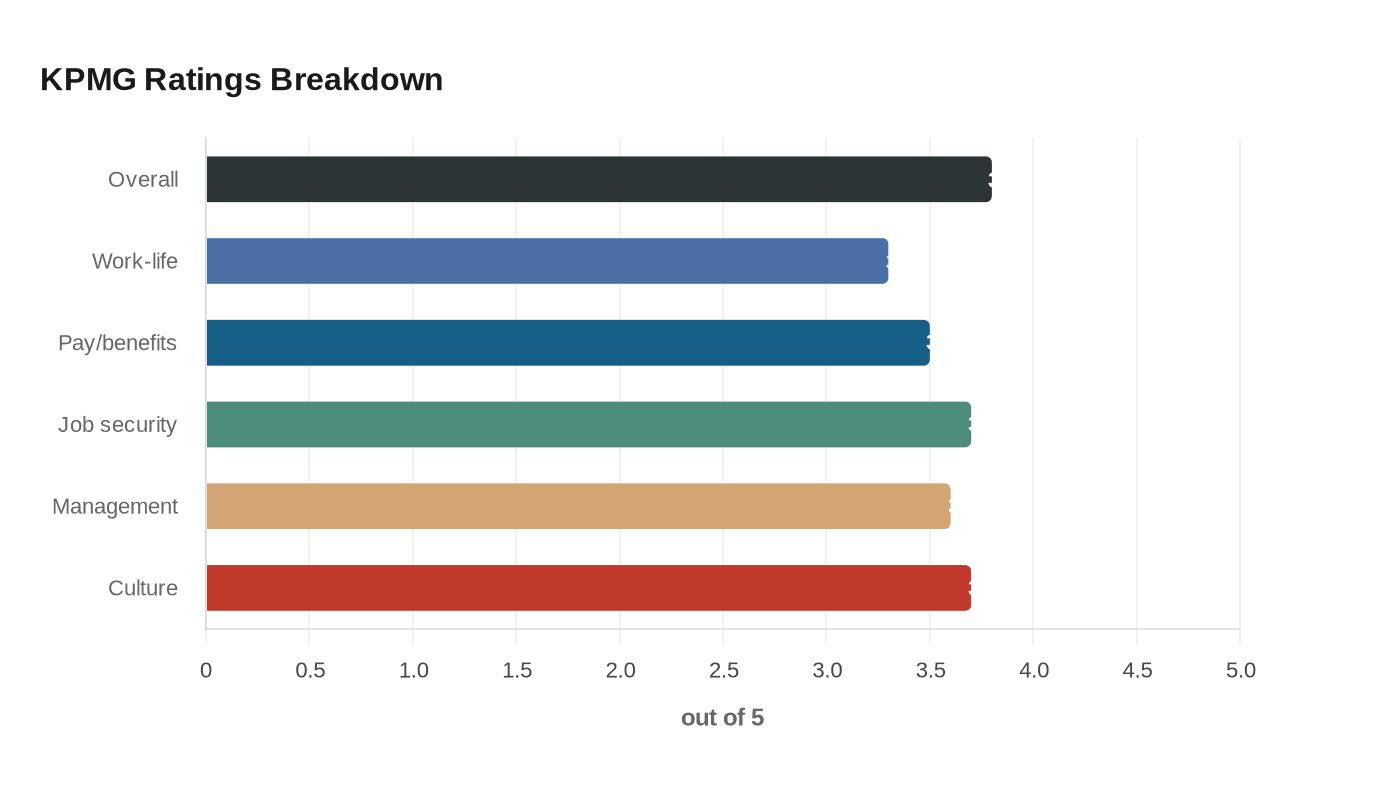
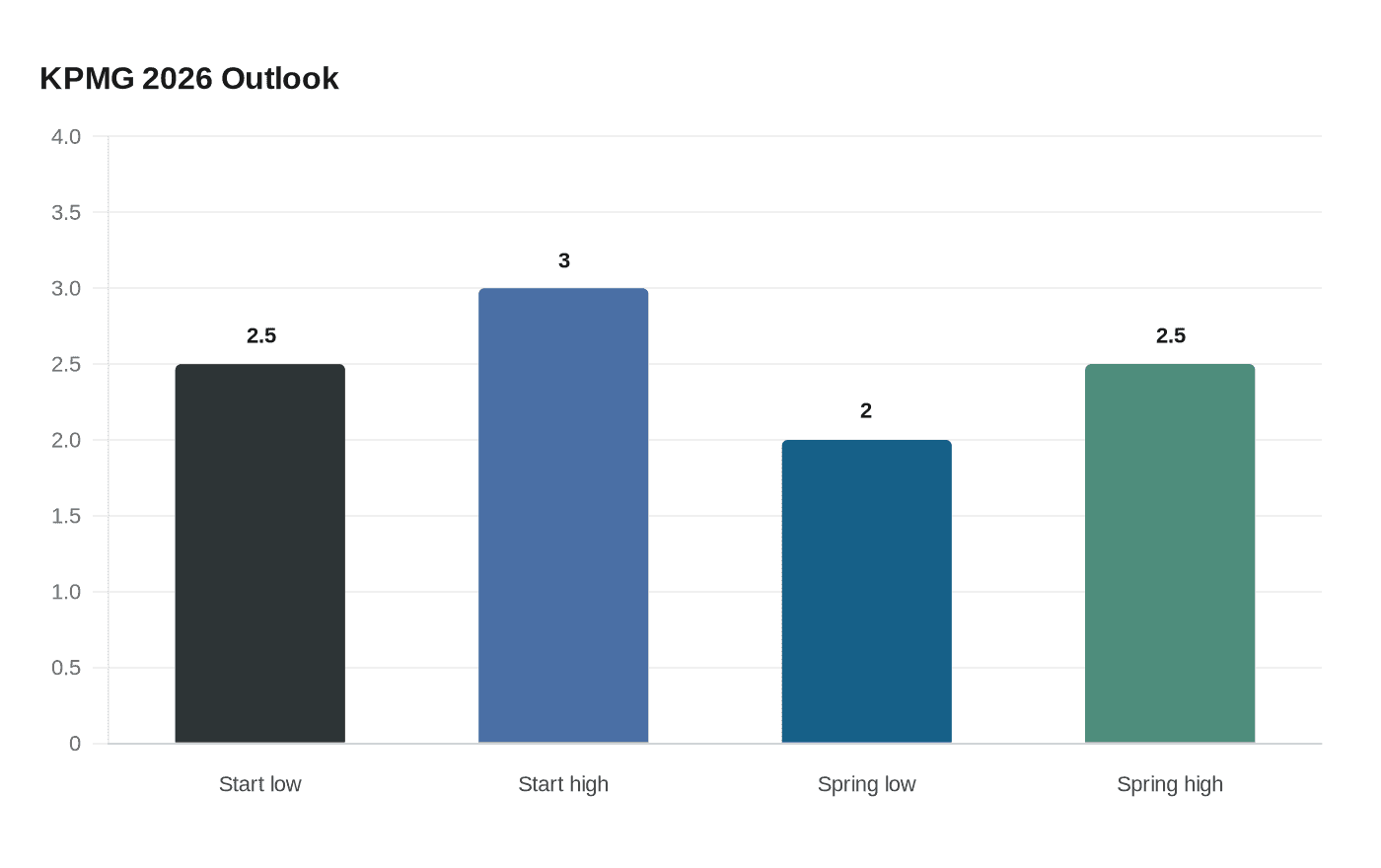KPMG flags Medicare reimbursement boost for AI healthcare tools, prior authorization proposal
CMS opened a reimbursement lane for AI care tools and pushed electronic prior auth into Medicare, Medicaid and ACA plans. Revenue cycle and compliance teams will feel it first.
By Monday morning, revenue cycle leaders, compliance officers and product teams will all be staring at the same question: which policy shift forces the most rework first?
The answer may be the prior-authorization proposal. CMS proposed mandatory electronic prior authorization for prescription drugs on April 10, and the Federal Register published it on April 14. The rule would reach Medicare Advantage, Medicaid fee-for-service, Medicaid managed care, CHIP fee-for-service and managed care, federally facilitated exchange Qualified Health Plan issuers, and some small-group SHOP plans. CMS says the goal is faster decisions, more transparency and less administrative burden, but for operators the work starts in the back office, where plan rules, data exchange and pharmacy workflows still collide with older systems.
That proposal lands at the same time CMS is trying to create a real reimbursement lane for AI and other technology-enabled care. The agency accepted more than 150 companies into ACCESS, the Advancing Chronic Care with Effective, Scalable Solutions model, a voluntary 10-year model that begins July 5, 2026. ACCESS is designed to support outcome-aligned payment in Original Medicare for technology-supported care options for chronic conditions, including high blood pressure, diabetes, chronic musculoskeletal pain and depression. Telehealth, wearables and apps are among the tools expected to fit inside that framework. For KPMG teams advising healthcare technology clients, that matters because reimbursement will decide which products stay pilots and which ones can scale into a usable care model.
The shortest path to operational friction is likely prior auth, but the longest tail may be drug pricing. Maryland’s Prescription Drug Affordability Board set an upper payment limit of $197 a month for Jardiance, far below the drug’s $573 list price, with the cap scheduled to take effect in January 2027. The state estimates the move could save state and local governments about $320,000 a year. It is only the second time a state board has imposed a drug price cap, which makes it a precedent other states and payer teams will be watching closely.
The transparency side is just as messy. A JAMA Network Open study of 2,464 Medicare hospitals found that institutions serving more low-income and minority patients were less likely to comply with hospital price transparency rules from early 2022 through late 2023. The gap points to a blunt operational reality: the hospitals most likely to be stretched on staffing and technology are also the ones most likely to lag on compliance.
The broader federal backdrop adds more churn. President Trump nominated Erica Schwartz, a retired Public Health Service rear admiral and former deputy surgeon general, to lead the CDC, and groups including the American Public Health Association praised her clinical and public-health background. For KPMG advisers, the immediate work is less about policy watching than implementation: reimbursement strategy for AI tools, prior-auth redesign, pricing analytics and the compliance systems that keep all of it moving.
Know something we missed? Have a correction or additional information?
Submit a Tip