Spent Coffee Grounds Remove Up to 98% of Heavy Metals From Water
Loughborough University researchers turned spent coffee grounds into biochar that pulled 98% of lead from water, trapping 4.9mg of lead per gram of material.

Researchers at Loughborough University published two studies showing that spent coffee grounds, processed into biochar or blended with rice husk, can strip lead, copper, and zinc from contaminated water with removal efficiencies reaching 98% for lead and more than 96% for copper and zinc in tested scenarios.
The first study, published in Biomass and Bioenergy, was led by Dr. Monika Mahajan and focused on converting spent grounds into a porous, carbon-rich biochar by subjecting them to high temperatures. The team optimized production conditions using a Box-Behnken experimental design and reported that the resulting biochar captured up to 98% of lead from water, with an adsorption capacity of 4.9 mg of lead per gram of biochar. "This work demonstrates how an everyday waste such as spent coffee grounds can be transformed into a high-value, sustainable adsorbent for removing toxic metals from water," Mahajan said. "By optimizing the decomposition conditions, we were able to significantly enhance the material's performance while keeping the process low-cost and environmentally friendly."
The second study, published in MDPI's Clean Technologies and led by Basmah Bushra, tested treated spent coffee grounds alone and blended with rice husk biochar inside a fixed-bed column targeting copper and zinc at low concentrations. Results showed that raw coffee waste performed better at low metal concentrations, specifically 2.5 ppm copper and 10 ppm zinc, while the coffee and rice husk blend pulled ahead at higher concentrations exceeding 5 ppm copper and 25 ppm zinc. Both formulations proved highly effective, with one test configuration removing 96% of the metals. "Our studies show that what we often dismiss as waste, like spent coffee grounds, can actually become powerful materials in tackling environmental pollution," Bushra said. "By turning waste into adsorption material, we can not only reduce landfill burdens but also create affordable materials for cleaning up contaminants."

The scale of potential feedstock is significant: global coffee consumption exceeded 176 million 60-kilogram bags in 2021-22, up from roughly 167 million bags the year prior, meaning the raw material for these adsorbents is generated in staggering quantities at cafes and roasteries every single day.
There is a technical wrinkle worth knowing about. A 2014 study published in the Journal of Environmental Management by Min-Suk Kim and colleagues found that when raw, uncharred spent coffee grounds were applied to acid mine drainage, heavy metal concentrations did decrease and pH increased, but phytotoxicity also increased because of a large release of dissolved organic carbon from the unprocessed material. The charred version, SCG-biochar, did not exhibit that problem. That distinction matters if these materials ever move beyond lab columns toward actual remediation sites where soil and plant life are factors.
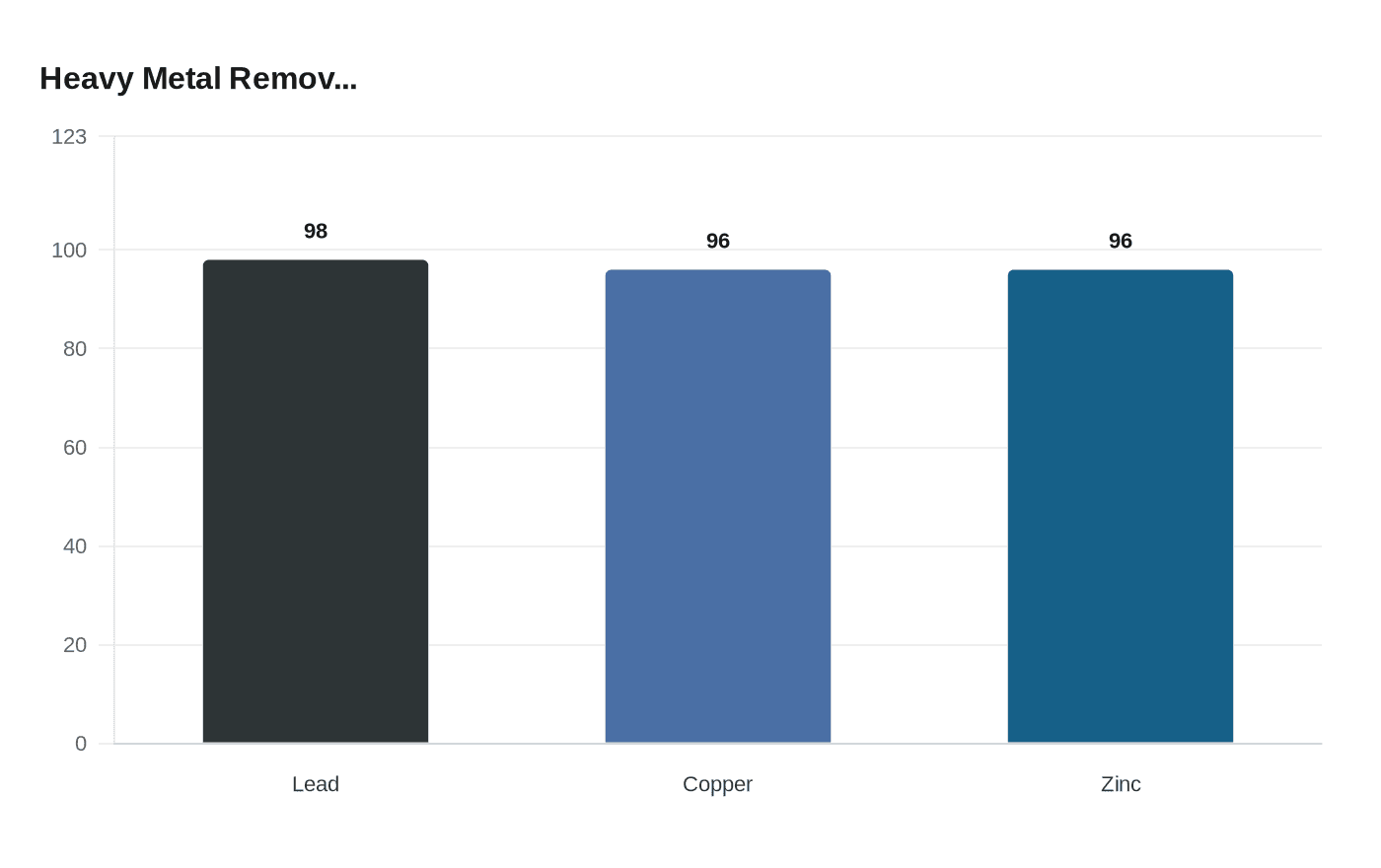
The Loughborough work frames the approach squarely within circular-economy thinking: coffee waste that currently heads to landfill becomes the filter that pulls toxic metals out of water. Whether that translates into deployable water treatment hardware will depend on further optimization and field testing, but 98% lead removal from a material most cafes pay to throw away is a number that tends to get attention.
Know something we missed? Have a correction or additional information?
Submit a Tip

