CDC Delays Report Showing COVID Vaccines Cut Hospitalizations by Half
Acting CDC Director Jay Bhattacharya blocked a completed agency report showing COVID vaccines halved hospitalizations for healthy adults, after it cleared internal scientific review.
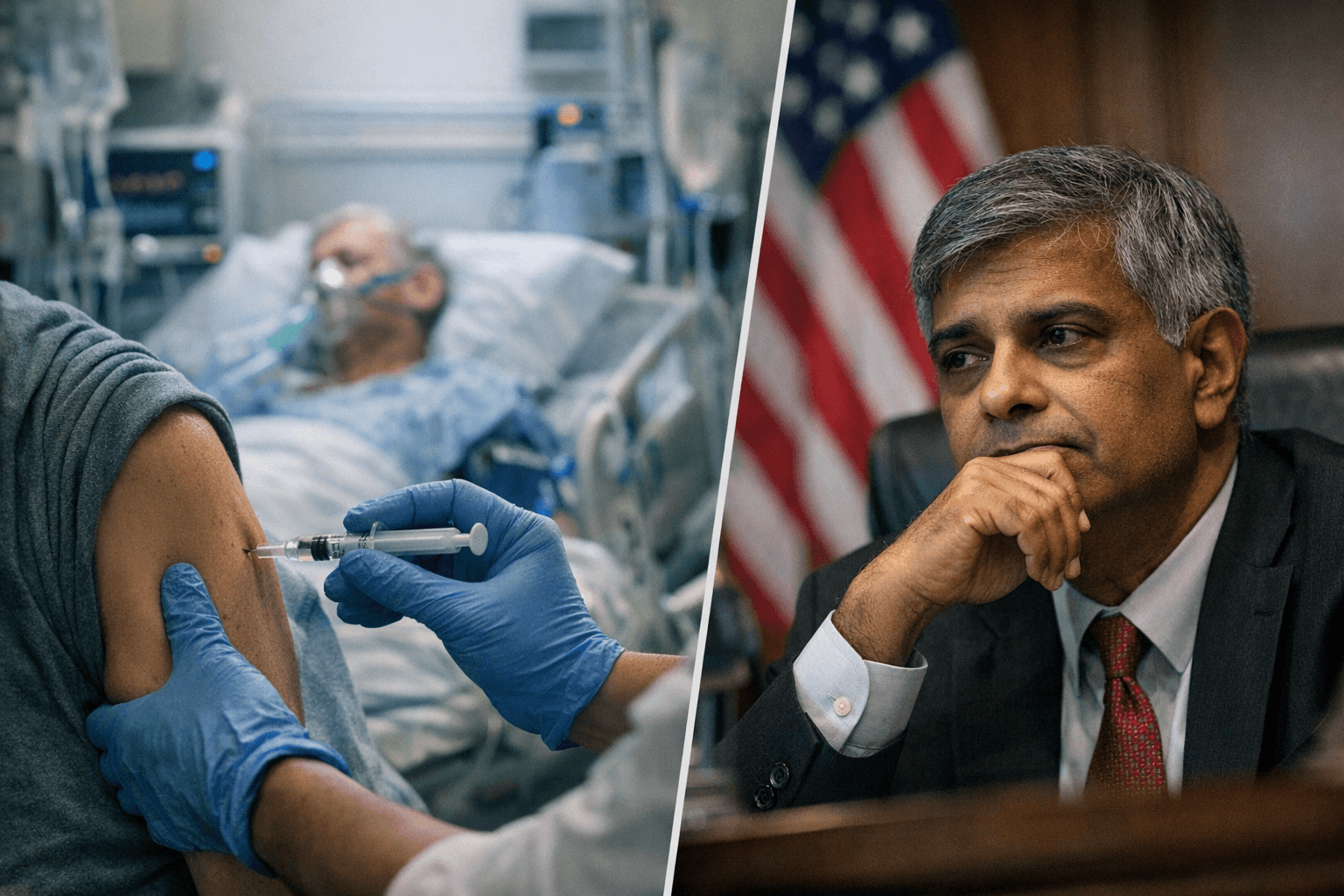
Acting CDC Director Jay Bhattacharya personally blocked publication of a completed agency report showing COVID-19 vaccines reduced emergency department visits and hospitalizations for healthy adults by approximately 50% last winter, according to two scientists with knowledge of the decision who spoke anonymously out of fear of retaliation.
The report had already cleared the CDC's own internal scientific-review process before Bhattacharya intervened. The delay has raised concerns among current and former officials that scientific findings conflicting with the priorities of HHS Secretary Robert F. Kennedy Jr. are being systematically sidelined.
Bhattacharya, whose full name is Jayanta Bhattacharya, cited concerns about the study's methodology as his stated reason for the hold. The methodology he questioned has been a standard CDC tool for years, used routinely to assess the effectiveness of flu shots and other respiratory virus vaccines. It also underpinned a landmark 2021 study published in the New England Journal of Medicine on COVID vaccine effectiveness, lending the approach substantial scientific credibility.
An HHS spokesperson, whose department oversees the CDC, described the situation as standard practice, saying it is "routine for CDC leadership to review" publications before release. A separate anonymous HHS official offered a more pointed defense, arguing the methodology is "prone to bias" because it relied on a hospitalized patient sample not representative of the general population. Bhattacharya was expected to meet with CDC scientists for further discussions before any final publication decision.
The delay landed against a politically charged backdrop inside the Trump administration. Some officials have been reluctant to push further on vaccine policy ahead of the midterm elections, while outside allies have been urging Kennedy to take a more aggressive posture in questioning vaccines. Some of Kennedy's handpicked vaccine advisors have pressed for stronger action. Kennedy, a longtime vaccine skeptic, has shaped a health policy environment in which agency researchers now worry that data pointing to vaccine benefits may not receive standard scientific treatment.
The 50% effectiveness finding carries direct public health weight. Emergency department visits and hospitalizations represent the most severe and costly outcomes of COVID-19 infection, and a vaccine effectiveness estimate of that magnitude for healthy adults would ordinarily move through CDC's established communications channels without leadership interference. That the report cleared internal review only to be stopped at the director's office marks a significant departure from the agency's normal process.
The two scientists who disclosed the delay did so only under anonymity, citing explicit concern about professional retaliation. That posture, rare for researchers at a federal public health institution, reflects the depth of unease inside the agency over how scientific findings are now being managed.
Know something we missed? Have a correction or additional information?
Submit a Tip

