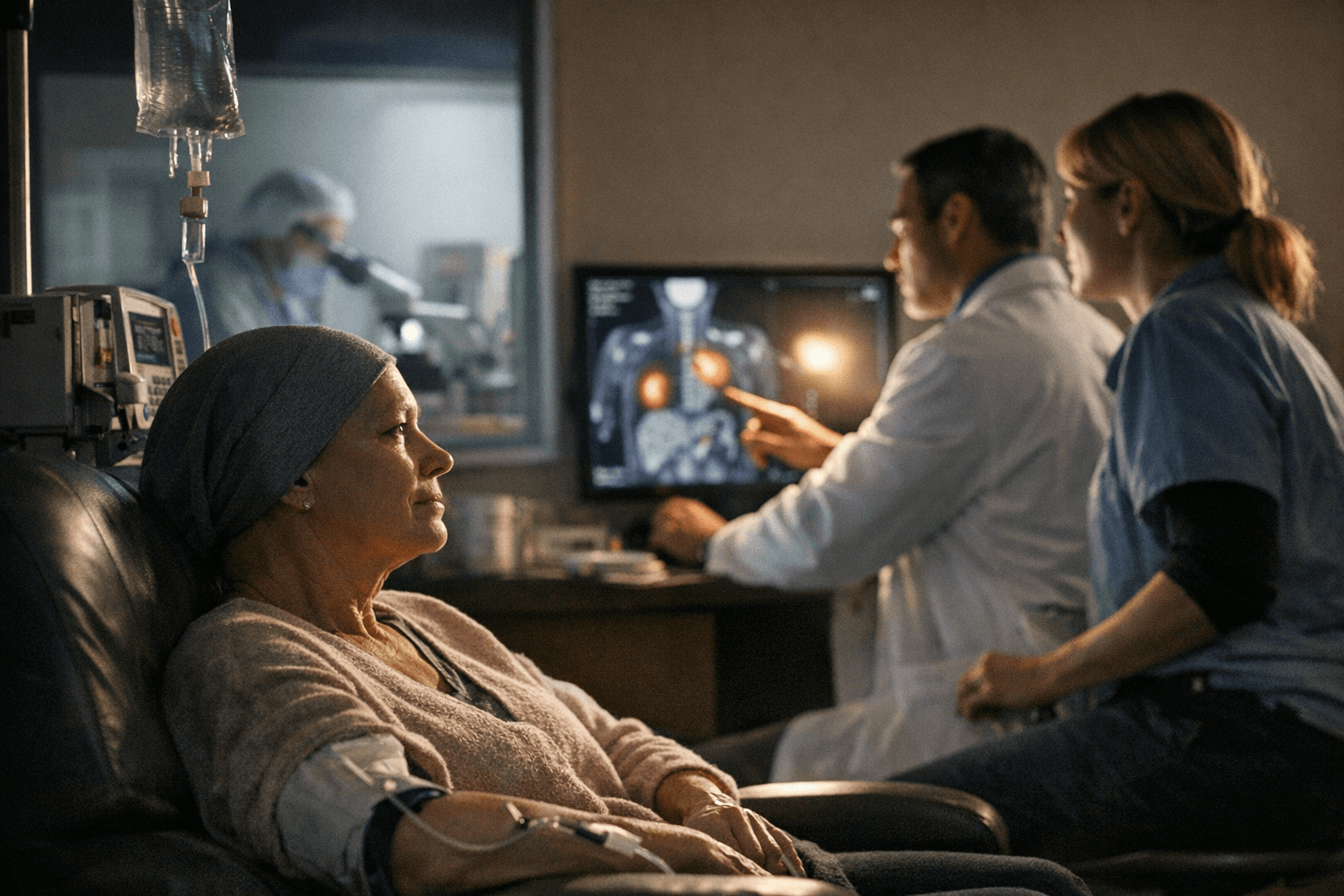
GSK says experimental cancer drug shows blockbuster potential in early trials
GSK’s Mo-Rez shrank tumors in 62% of platinum-resistant ovarian cancer patients, but the drug is still in phase 1 and years from proving it can change care.
GlaxoSmithKline’s experimental cancer drug Mo-Rez produced tumor shrinkage rates that caught the market’s attention, but the company is still a long way from proving it can alter treatment for some of the hardest gynecologic cancers.
In the BEHOLD-1 study, GSK said 62% of patients with platinum-resistant ovarian cancer and 67% of patients with recurrent or advanced endometrial cancer saw their tumors shrink by at least 30%. The data were presented in a late-breaking oral session at the Society of Gynecologic Oncology Annual Meeting on Women’s Cancer in San Juan, Puerto Rico.
For GSK, that is enough to call the drug one of its priority assets and to say it has blockbuster potential. In biotech, blockbuster usually means a medicine that can generate more than $1 billion in annual sales. That is a commercial benchmark, not a guarantee of medical success, and Mo-Rez is still only in early testing.
The drug, also known as mocertatug rezetecan and previously referred to as HS-20089 and GSK5733584, is an antibody-drug conjugate that targets B7-H4, a protein GSK says is widely expressed in ovarian and endometrial cancers but low in normal tissue. The company said responses appeared across a range of B7-H4 expression levels, a sign the treatment may not be limited to a narrow biomarker-defined group.
Hesham Abdullah, who leads oncology research at GSK, has pointed to the company’s accelerated pace in cancer development under chief executive Luke Miels. GSK named Miels as CEO designate on September 29, 2025, and he took full chief executive responsibilities and joined the board on January 1, 2026.
The promise comes against a tough backdrop. GSK says treatment options remain limited for platinum-resistant ovarian cancer and advanced endometrial cancer, two settings where new therapies have struggled to deliver durable benefit. Company and congress materials also said nausea was the most common treatment-related adverse event in BEHOLD-1, underscoring that safety will matter as much as response rates in later studies.
GSK is already running two late-stage trials and said it plans to launch five pivotal global phase III trials in 2026, including BEHOLD-Ovarian02, BEHOLD-Ovarian03 and BEHOLD-Endometrial02. Those studies will have to show not just that tumors shrink, but that patients live longer, stay stable longer and tolerate treatment well enough for it to matter in routine practice.
The company licensed the drug from Hansoh Pharma in October 2023, taking exclusive worldwide rights outside mainland China, Hong Kong, Macau and Taiwan. That deal gives GSK a shot at building a larger oncology franchise around B7-H4, but the distance from early response data to standard care remains substantial.
Know something we missed? Have a correction or additional information?
Submit a Tip