Scientists Identify GPR133 Receptor as Key Target for Stronger Bones
AP503, a small-molecule compound, boosted bone density in mice by activating GPR133, pointing scientists toward a new osteoporosis target; human trials remain years away.

Researchers have identified GPR133, a receptor previously unconnected to bone metabolism, as a powerful regulator of skeletal strength. In mice treated with a small-molecule compound called AP503, scientists measured statistically significant improvements in bone mineral density, microarchitectural bone quality and mechanical resistance to fracture-like stress compared with untreated controls, raising the possibility of a new therapeutic class for age-related bone loss.
The findings arrive against a backdrop of substantial unmet medical need. Osteoporosis and related fractures cause widespread disability, mortality and healthcare costs, particularly among older adults, and existing drug options leave a meaningful share of patients either inadequately treated or unable to tolerate the available therapies.
Current treatments divide into two broad categories: agents that slow bone breakdown, including bisphosphonates and the biologic denosumab, and anabolic drugs that stimulate new bone formation, such as teriparatide. Each carries constraints. Bisphosphonates can lose efficacy over time, and anabolic agents require injectable administration with limits on treatment duration. GPR133 activation offers a mechanistically distinct possibility: driving osteoblast activity, the cells responsible for building bone, with a signaling profile researchers argue produces less of the resorption interference that complicates several existing drugs. The precise pathways through which GPR133 exerts that effect will require further elucidation.
The laboratory results, however promising, mark only the beginning of a lengthy development pipeline. Advancing AP503 toward patients would first require dose-finding toxicology work, then Phase 1 safety trials in healthy volunteers, followed by Phase 2 proof-of-concept studies in patients with confirmed low bone density or osteoporosis. Larger pivotal trials would ultimately need to demonstrate actual reductions in fracture rates over extended follow-up periods before any regulatory body could consider approval.
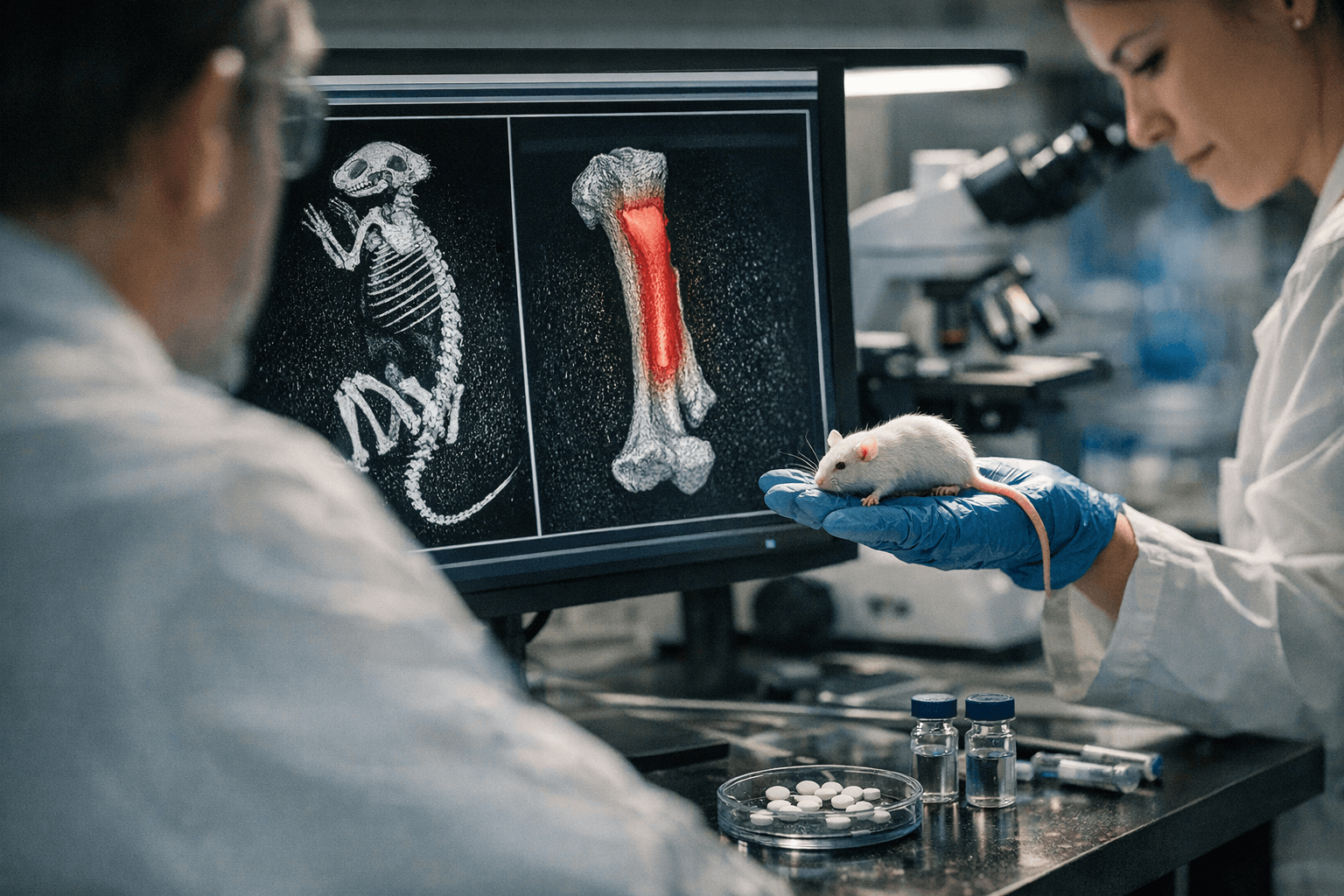
Human biology adds layers of complexity absent from mouse models. Hormonal, metabolic and genetic variables unique to patients could alter responsiveness to a GPR133-targeted drug, and the compound's off-target effects would need mapping across multiple organ systems. Scientists cautioned that preclinical success in bone research has frequently failed to translate into human benefit, a pattern that argues for cautious rather than accelerated progression through the development stages.
If that pipeline eventually produces a viable drug, a GPR133-targeted therapy could join or supplement bisphosphonates, denosumab and anabolic agents, expanding options precisely for the patients current regimens fail. The identification of GPR133 as a bone-regulating target and AP503 as its activator represents an early but notable step in that process, one that gives researchers a defined molecular address to pursue in the years of testing ahead.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

