Scientists Race for Hantavirus Treatments as Deadly Cases Persist
Hantavirus has killed 35% of U.S. cases, yet no licensed treatment exists as scientists push vaccine candidates from mouse studies to early trials.

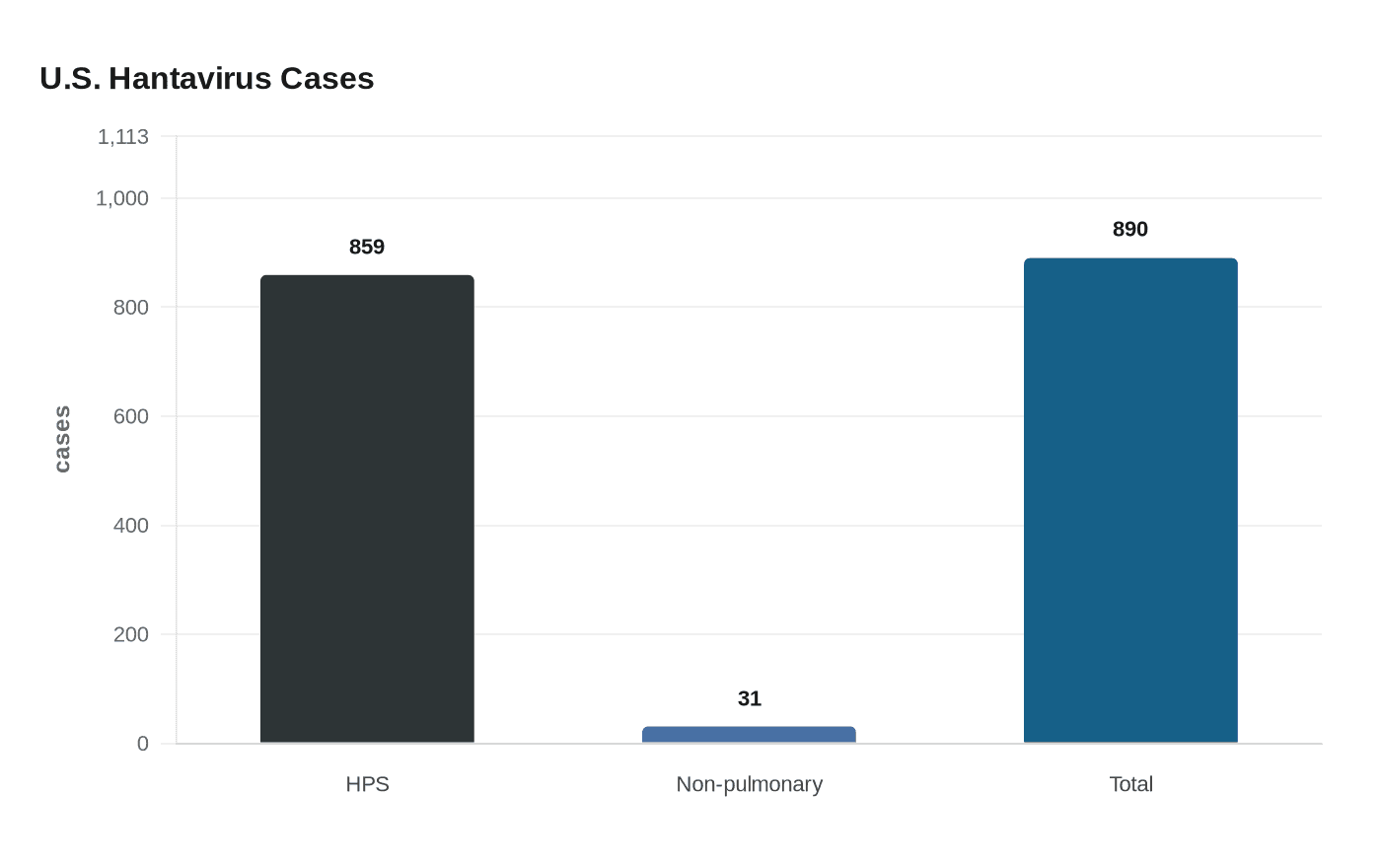
Hantavirus has killed 35% of the 890 laboratory-confirmed cases recorded in the United States through the end of 2023, and the disease still has no specific treatment beyond supportive care. The Centers for Disease Control and Prevention says 859 of those infections were hantavirus pulmonary syndrome and 31 were non-pulmonary infections, with 94% of reported U.S. cases occurring west of the Mississippi River.
The pattern has changed little since surveillance began in 1993 after the Four Corners outbreak in Arizona, Colorado, New Mexico and Utah. Hantavirus pulmonary syndrome became nationally notifiable in 1995, but cases remain sporadic, usually in rural areas, where the deer mouse is the primary host in western states. The virus spreads when people inhale aerosolized urine, feces or saliva from infected rodents. The CDC says Andes virus is the only hantavirus known to spread person-to-person.
That public-health reality has kept the drug and vaccine pipeline thin. The World Health Organization says there is no licensed specific antiviral treatment or vaccine for hantavirus infection, and that care focuses on close monitoring and treatment of respiratory, cardiac and kidney complications. Severe patients may require ICU-level breathing support or intubation, which is why early referral to a facility with intensive-care capability can improve survival. Even with those stakes, scientists say hantaviruses remain underfunded compared with better-known threats, in part because outbreaks have not sustained the national attention that typically drives commercial investment and regulatory momentum.
Some of the most promising work is finally moving forward. A University of Texas at Austin team led by Jason McLellan mapped the Andes virus Gn-Gc tetramer at the highest resolution yet and used the structure to produce a vaccine candidate that triggered neutralizing antibodies in mice. The finding matters because it points to both vaccine design and antibody therapy for a family of viruses with no approved countermeasures.
Other efforts are advancing more slowly. A 2024 phase 1 clinical trial tested Hantaan and Puumala virus DNA vaccines delivered by intramuscular electroporation, while preclinical programs are exploring protein subunit, virus-vectored, nucleic-acid and recombinant VSV-based approaches. Researchers say those efforts show the field is no longer empty, but a publicly available shot is still likely years away without sustained federal support. For hantavirus, the central question remains whether investment arrives before the next outbreak forces the issue again.
Know something we missed? Have a correction or additional information?
Submit a Tip

