Two treatments show promise against deadly pancreatic cancer
Two experimental pancreatic cancer treatments extended survival in early studies, including an oral KRAS pill that doubled life expectancy against standard chemotherapy.
Two experimental treatments have added rare momentum in pancreatic cancer, a disease that remains the third leading cause of cancer-related death in the United States and still carries a five-year relative survival rate of about 13 percent overall. In distant-stage disease, survival falls to about 3 percent, and roughly half to 55 percent of patients are already metastatic when diagnosed.
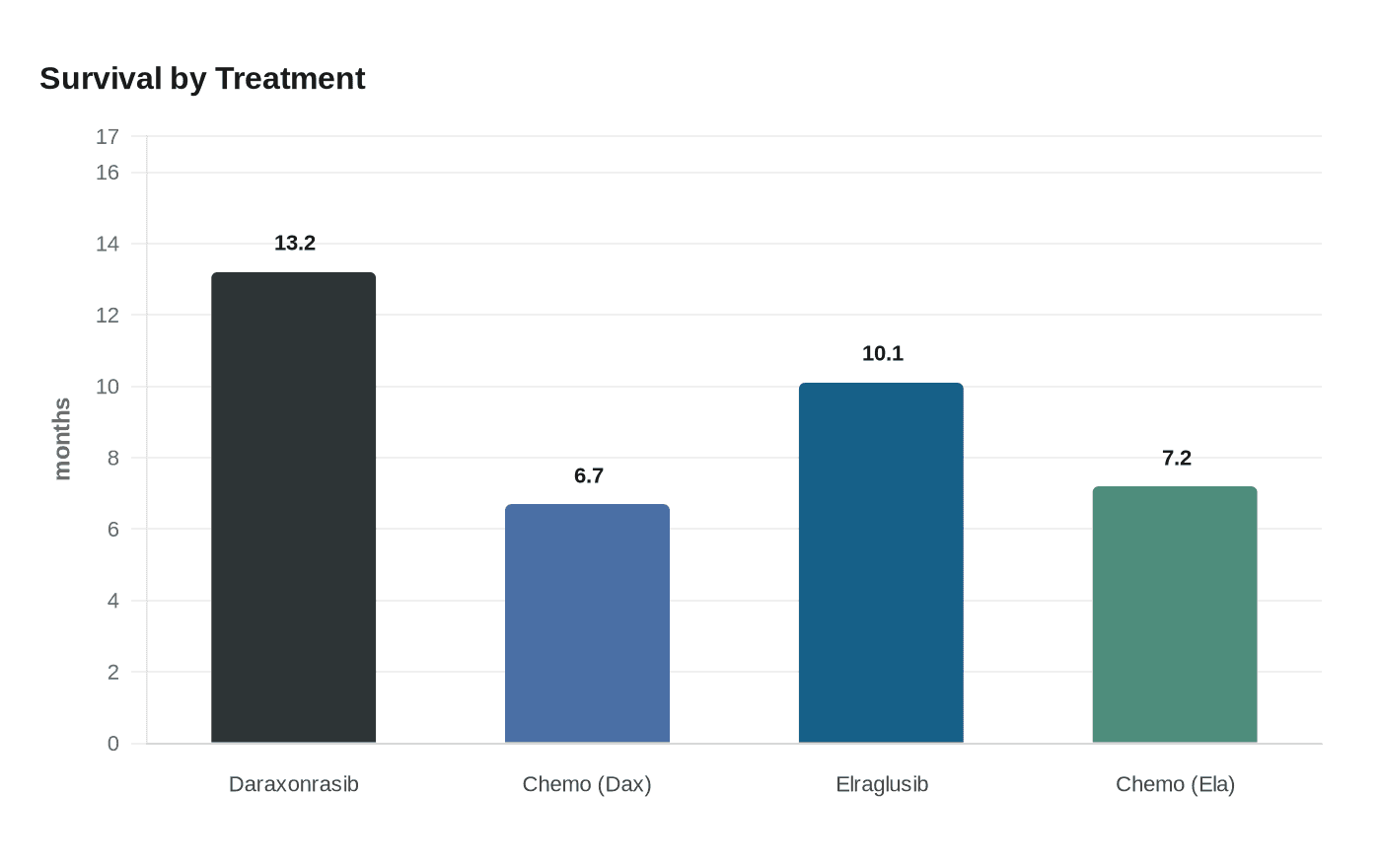
One of the most closely watched signals came from daraxonrasib, an oral pill from Revolution Medicines that targets KRAS, the mutation that drives more than 90 percent of pancreatic cancers. In a Phase III trial, patients lived an average of 13.2 months after starting the drug, compared with 6.7 months for those who received standard chemotherapy. The company said it plans to seek approval from the U.S. Food and Drug Administration, and the agency has already granted daraxonrasib a Commissioner’s National Priority Voucher, a designation intended to speed review.
Eileen O’Reilly, a gastrointestinal medical oncologist at Memorial Sloan Kettering Cancer Center who led one of the studies, said the findings could mark a turning point. She called them a "landmark shift" and said they may help build targeted therapy into the backbone of pancreatic cancer treatment. O’Reilly also said the results "hopefully set the stage for building on targeted therapy as a major backbone for the treatment of pancreas cancer, and a key goal now is to build and extend these results in all stages."
A second study pointed to another possible path. Elraglusib, an intravenous drug developed by Northwestern researchers and supported by Actuate Therapeutics, was tested in a randomized Phase II trial of 233 patients with metastatic pancreatic cancer across 60 sites in six countries in North America and Europe. Patients who received elraglusib plus standard chemotherapy were twice as likely to be alive after one year as those who received chemotherapy alone, and the drug cut the risk of death by 38 percent. Median overall survival was 10.1 months, versus 7.2 months with chemotherapy alone. The findings were published in Nature Medicine on April 14, 2026.
Devalingam Mahalingam, the lead author, said the results offer "cautious optimism" and stand out because they are among only a few randomized trials in the past decade to show a survival benefit that may apply broadly across pancreatic cancer patients. Taken together, the two studies, highlighted at the American Association for Cancer Research annual meeting in San Diego from April 17 to April 22, showed how researchers are still pressing ahead with targeted therapy and chemotherapy combinations in a field that has long seen too little progress.
Know something we missed? Have a correction or additional information?
Submit a Tip