GLP-1 boom drives surge of interest in peptide therapies
Andrew Huberman says GLP drugs have pulled peptides into the mainstream, as Wegovy and Zepbound turn a niche science into a multibillion-dollar market.
Andrew Huberman’s latest comments landed inside a bigger market shift: GLP-1 drugs have pushed peptides out of the research fringe and into mainstream weight-loss and cardiometabolic care. What started as a diabetes story now drives obesity medicine, investor attention and a growing public appetite for anything framed as a protein-derived therapy.
Peptides are short chains of amino acids, smaller than full proteins, and in medicine they can be designed to act with precision on specific receptors. That is part of their appeal in the GLP-1 era. Semaglutide, sold as Wegovy for obesity care, won FDA approval for chronic weight management on June 4, 2021, and the agency later expanded the label on March 8, 2024, to reduce the risk of cardiovascular death, heart attack and stroke in adults with established cardiovascular disease and overweight or obesity. That broader use helped turn peptide therapeutics into a public-health and commercial story, not just a sports or longevity trend.

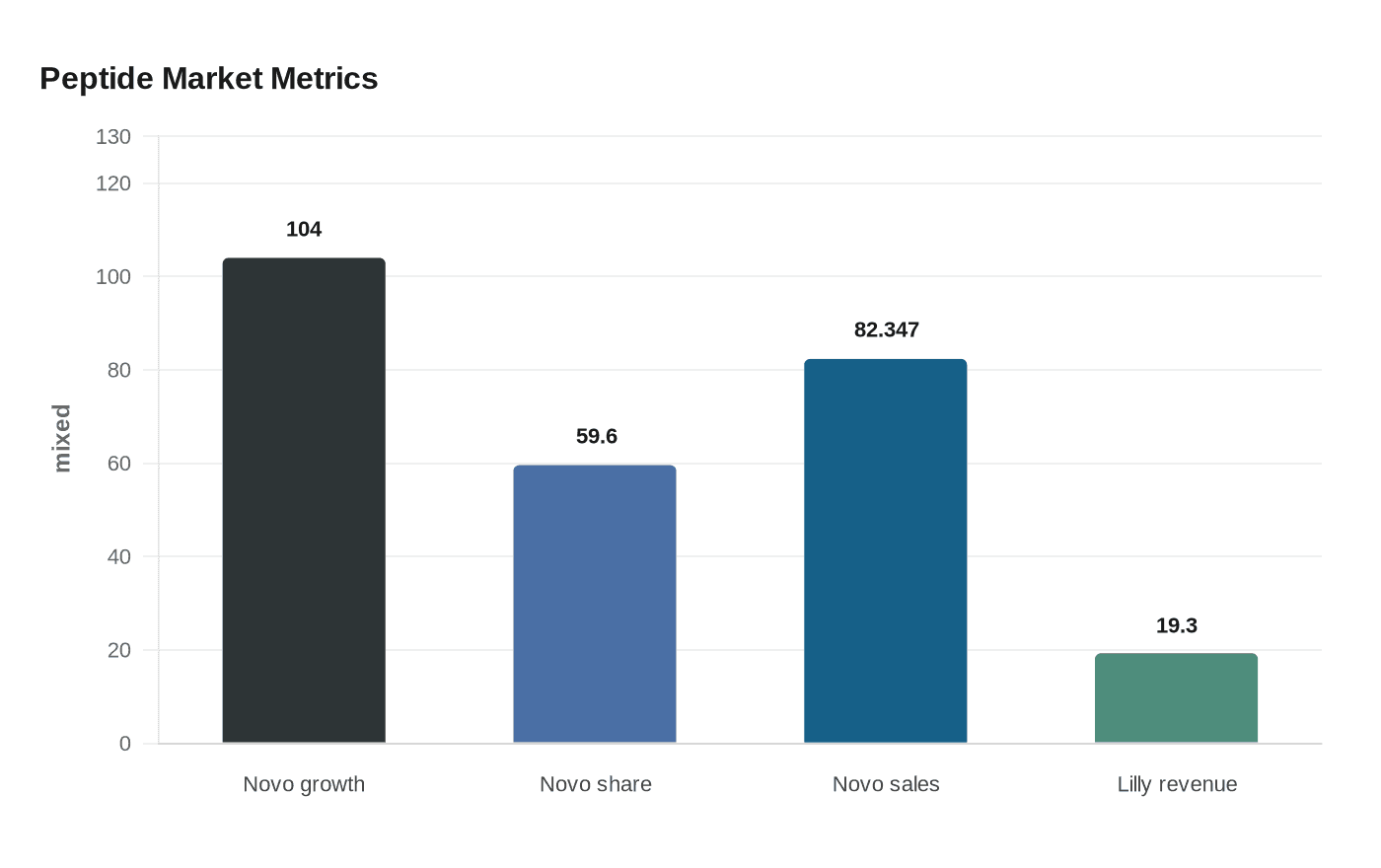
The scale of the market is now hard to miss. Novo Nordisk said in 2025 that global branded GLP-1 obesity market volume grew 104 percent and that it held 59.6 percent branded volume market share. The company also said obesity care sales reached DKK 82.347 billion. Eli Lilly and Company reported fourth-quarter 2025 revenue of $19.3 billion, up 43 percent from a year earlier, with Mounjaro and Zepbound doing the heavy lifting. For companies in Bagsværd, Denmark, and Indianapolis, Indiana, peptide drugs are no longer a side category. They are the center of the business.
The clinical case for the class is anchored by hard outcomes, not hype. Wegovy’s cardiovascular-risk approval rested on the SELECT trial, which enrolled about 17,500 patients with established cardiovascular disease and overweight or obesity. That evidence is one reason the GLP-1 story has been so durable, even as demand has strained supply and sparked imitation products.
The caution flags are just as real. On April 1, 2026, the FDA said the tirzepatide shortage had been resolved and reminded compounders of the rules that apply as national GLP-1 supply stabilizes. Before that, FDA warnings highlighted concerns about compounded GLP-1 products and active pharmaceutical ingredients imported from unverified foreign sources. Huberman has been discussing peptide therapeutics for more than a year, including a Huberman Lab episode on the benefits and risks of peptide therapeutics in April 2024 and a March 9, 2026 interview on the emerging peptide and GLP landscape. The message in that arc is clear: the legitimate science is real, but the boom also invites overreach, and regulators are still chasing the gray zones.
Know something we missed? Have a correction or additional information?
Submit a Tip

