Stanford researchers map blood proteins to reveal accelerated organ aging
Stanford’s blood-protein map shows one in five older adults may have at least one organ aging fast, but the therapeutic leap is still mostly in mice and tissue samples.

Stanford researchers are turning the promise of “rejuvenation proteins” into something more practical: a blood-based map that can flag which organs are aging out of step long before symptoms appear. The translational reality is still important, though. The strongest evidence now comes from plasma signatures in people, brain and cartilage rescue experiments in animals and tissue samples, and only early-stage drug development on the treatment side.
The Stanford-led study, published in Nature on December 6, 2023, analyzed nearly 5,000 plasma proteins from 5,676 adults and built machine-learning aging models for 11 organs and tissues: heart, fat, lung, immune system, kidney, liver, muscle, pancreas, brain, vasculature and intestine. Almost 900 of the proteins were enriched in a single organ, giving Tony Wyss-Coray’s group a way to separate organ-specific wear and tear from a single broad biological-age score.
The numbers were striking. Stanford Medicine said 18.4% of adults age 50 and older had at least one organ aging significantly faster than average, while only about 1 in 60 had two organs aging at that pace. Across the population, nearly 20% showed strongly accelerated aging in one organ and 1.7% were multi-organ agers. That pattern mattered clinically: accelerated organ aging tracked with a 20% to 50% higher risk of death, and accelerated heart aging carried a 250% higher risk of heart failure. Brain and vascular aging also predicted Alzheimer’s disease progression as strongly as plasma pTau-181.
The work fits a broader shift in aging research away from one catch-all measure of “biological age” and toward organ-by-organ clocks that can guide earlier intervention. NIH summarized the same study by noting that people with hypertension and diabetes tended to have older kidneys, while older hearts lined up with atrial fibrillation and heart attacks. In other words, the readout is not just academic. It points to where disease may emerge first.
That diagnostic map is now being matched by a handful of mechanistic leads. A March 21, 2024 summary from the National Institute on Aging highlighted platelet factor 4, or PF4, a blood-clotting protein elevated in young mice that reduced brain inflammation and improved cognition in old mice in work involving Stanford, UCSF and Duke University. More recently, Stanford Medicine reported in November 2025 that blocking 15-PGDH, described as a master regulator of aging, reversed cartilage loss in mice and triggered new functional cartilage in human knee tissue from replacement surgeries. The oral version of that treatment was already in clinical trials for age-related muscle weakness.
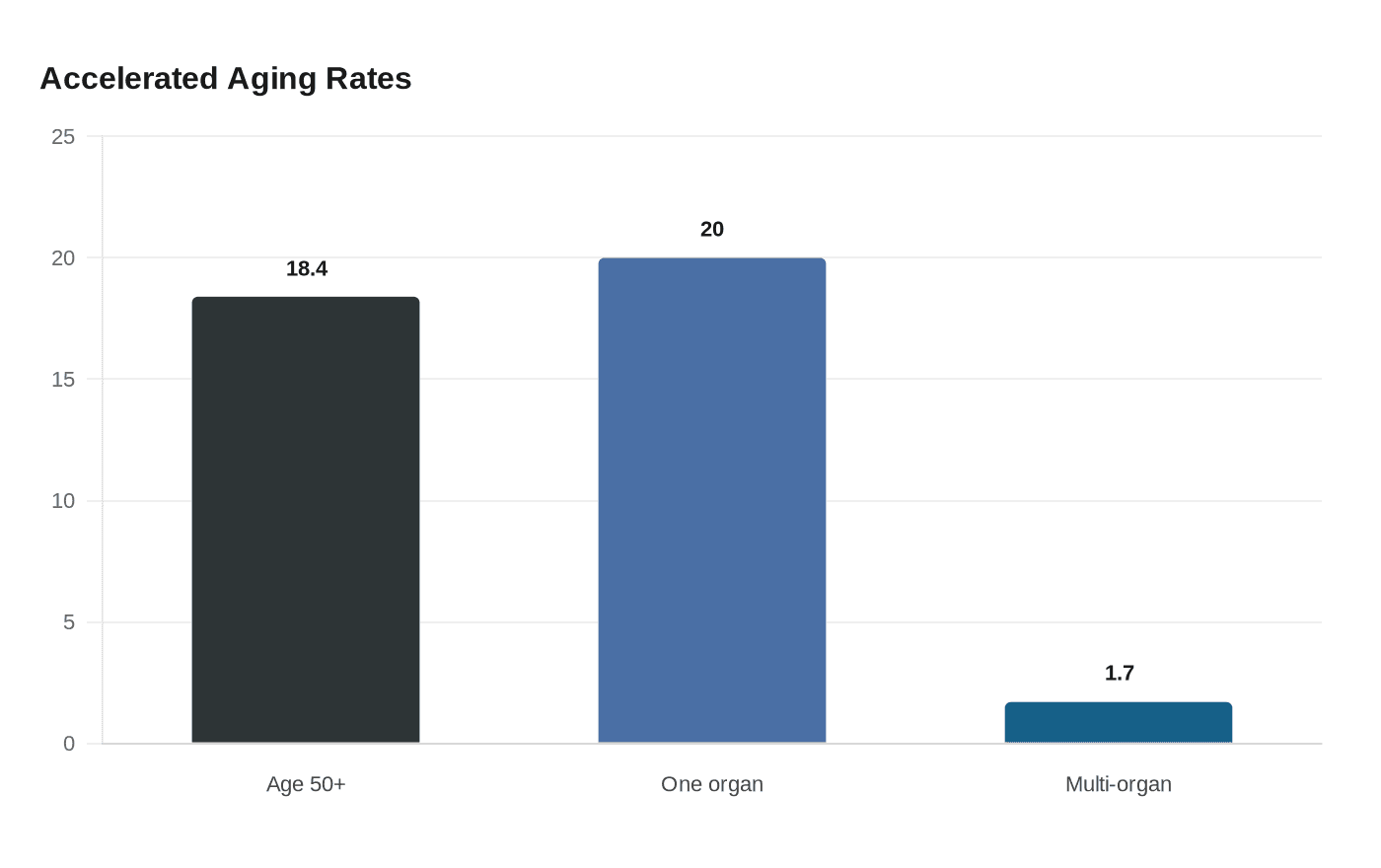
That is the commercial question hanging over the field. Blood-protein mapping is moving quickly into clinical relevance, but the delivery challenge remains substantial: turning circulating signals into organ-targeted, durable therapies that work in humans. For now, Stanford’s results look less like a finished anti-aging treatment and more like the blueprint for the next generation of regenerative medicine.
Know something we missed? Have a correction or additional information?
Submit a Tip

