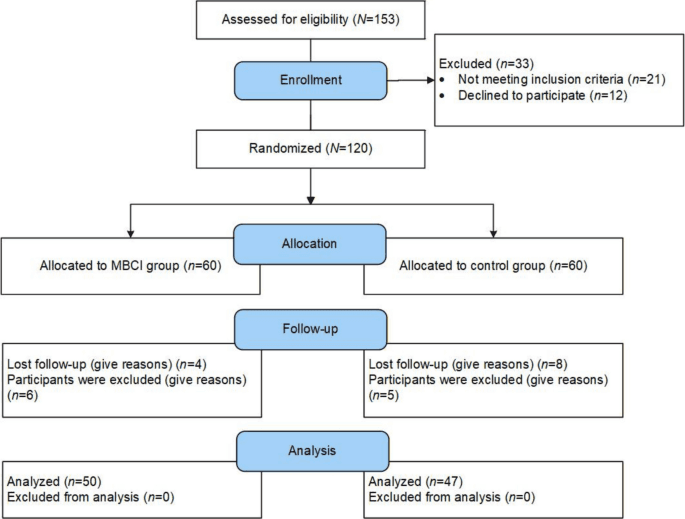
Randomized trial registers mindfulness-integrated childbirth education targeting fear, prenatal stress, self-efficacy
ClinicalTrials.gov entry NCT07415122 was updated Feb 11, 2026 to register a randomized trial testing mindfulness-integrated childbirth education on fear of childbirth, prenatal stress and childbirth self-efficacy.

ClinicalTrials.gov record NCT07415122 was updated on February 11, 2026 to register a randomized controlled trial that evaluates the effect of mindfulness-integrated childbirth education on fear of childbirth, prenatal stress, and childbirth self-efficacy. The public excerpt in the registry is truncated and includes the literal fragment “participants are randomized to childbirth education wi,” with no further procedural details in the available text.
Key design elements are stated in the registry entry only as a randomized controlled trial and the three named outcomes; several operational details are absent from the excerpt. The update supplies no sample size, no study sites or locations, no principal investigator or institutional affiliation, and no intervention schedule, curriculum or measurement instruments, items that will need to be pulled from the full ClinicalTrials.gov record or the study protocol to assess dose, comparator definition and planned analyses.
The new childbirth education registration arrives amid a wave of recent mindfulness trials that show a mix of preliminary and peer‑reviewed findings. University of California Health listings updated February 2, 2026 catalog 17 mindfulness clinical trials in progress and 11 that are open to eligible people, and name lead scientists including Andrew Fuligni, Ph.D., Sean D Young, Eric Garland, PhD, Karen L Lindsay, RD, PhD, Nisha Acharya, MD, MS, Lauren E Gyllenhammer, PhD, Jason Satterfield, PhD, Wolf E Mehling, MD, and Jacqueline H. Kim.
Randomized evidence from other registries illustrates trial models the childbirth study may draw on. The Chinese Clinical Trial Registry entry ChiCTR2400088029 registered August 9, 2024 describes a Mindfulness‑Based Auditory Hallucination Management (MBAHM) randomized trial in schizophrenia with 80 patients randomly assigned to MBAHM (n = 40) or control (n = 40). The MBAHM group received “a 2-h MBAHM intervention twice a week for eight weeks,” and the reported outcomes showed significant reductions in auditory hallucinations, anxiety and depression and improvements in quality of life compared to baseline and control, with p < 0.001 and correlation analyses linking symptom reductions to quality-of-life gains.
Digital mindfulness delivery is also advancing to phase III. NRG-CC015, titled “Harnessing E-Mindfulness Approaches for Living After Breast Cancer - HEAL-ABC,” is a prospective, randomized phase III trial comparing a live instructor‑led Zoom program (MAPs LO) to an app‑based self‑paced program (MAPs App). The trial targets younger breast cancer survivors diagnosed at or before age 50 who completed primary treatment at least six months earlier and report elevated depressive symptoms; UC Irvine is listed as a site and UC BRAID notes the trial is “open to eligible people ages 18-50.”
App-based, daily practice formats have already produced peer-reviewed results. The University of Tokyo randomized controlled trial registered UMIN000051796 ran from July 2023 to January 2024; participants in the intervention group installed a meditation app and practiced guided mindfulness meditation on their phones every day for eight weeks. Published February 2, 2026 in JMIR Vol 28, the study concluded the app reduced perceived stress, depressive and anxiety symptoms and trait anger and improved life satisfaction, while noting higher-intensity interventions may be needed to address work- and family-related indicators.
Smaller program evaluations point to feasibility and data-handling practices investigators are adopting. A Cardio JMIR study registered UMIN000044075 reported that a mindfulness program for pulmonary hypertension appeared to support symptom and emotion management and suggested potential improvements in quality of life; the paper notes data were anonymized, stored on a password-protected hard disk drive and that participants were not paid but received the program free of charge.
Next reporting steps to clarify the childbirth trial: obtain the full ClinicalTrials.gov entry for NCT07415122 to extract sponsor, principal investigator, planned enrollment, inclusion criteria, intervention dose and comparator details, measurement instruments and timelines; request the ChiCTR record or manuscript for ChiCTR2400088029 to confirm investigator names and instruments; and pull NRG-CC015 protocol information for sample size and endpoints. Which of those records would you like me to retrieve first?
Know something we missed? Have a correction or additional information?
Submit a Tip