Short mindfulness trial targets postpartum anxiety with brain imaging
A protocol outlines a 4-week mindfulness trial for postpartum anxiety and plans resting-state fMRI to study neural change. This could signal scalable, nonpharmacologic options for new parents.
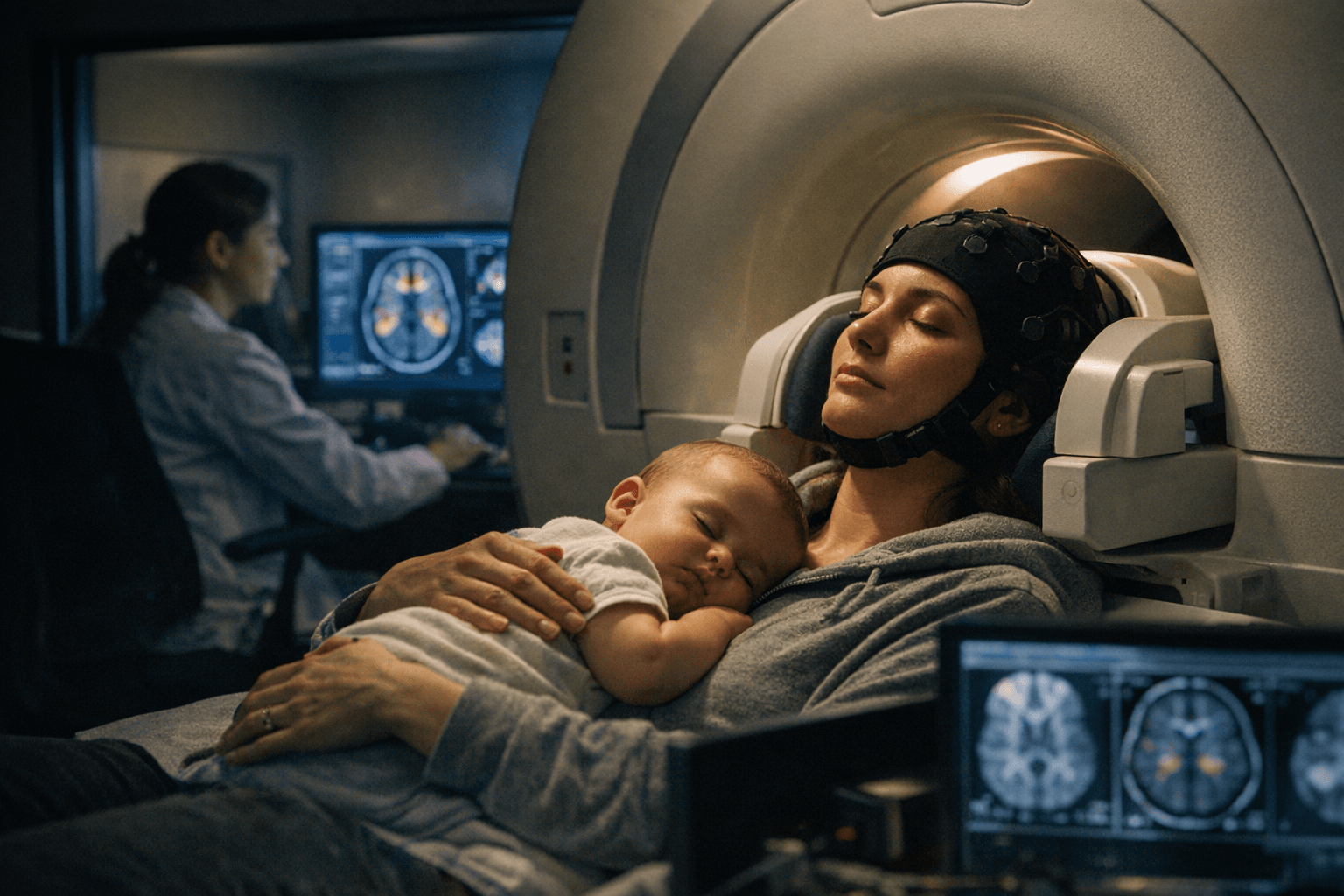
Researchers published a detailed protocol on January 13, 2026, for a proof-of-concept randomized controlled trial testing a brief, 4-week mindfulness-based intervention aimed at anxiety disorders in the postpartum period. The protocol lays out the study aims and hypotheses, participant inclusion and exclusion criteria, recruitment strategies, the intervention curriculum tailored for postpartum parents, comparator conditions, and the outcome measures that will determine feasibility and early efficacy.
The intervention is deliberately brief and scalable, designed to fit the time constraints and fluctuating schedules of new parents. Content centers on mindfulness practices adapted for postpartum life, with explicit components to strengthen emotion regulation and interoception. Those mechanistic targets informed the choice of outcome measures: primary outcomes include validated anxiety scales and established measures of emotion regulation, assessed before and after the intervention.
To explore neural mechanisms, the protocol includes resting-state fMRI scans pre- and post-intervention. The imaging analysis will focus on changes in connectivity within the default mode network and the salience network, networks implicated in self-referential processing, attentional shifts, and the detection of salient internal and external signals. Including neuroimaging in a short pilot seeks to link clinical signals to measurable brain changes even in a compact, parent-friendly format.
The protocol also defines recruitment plans and comparator conditions so the trial can evaluate not just clinical signal but feasibility metrics such as enrollment rates, session adherence, retention, and safety. Ethics and safety procedures are described alongside planned statistical analyses appropriate for a pilot study, with the authors emphasizing that the trial is intended to test feasibility and gather preliminary estimates rather than provide definitive efficacy claims.
For the mindfulness community and practitioners working with perinatal populations, this protocol matters because it prioritizes accessibility without sacrificing mechanistic clarity. If the pilot demonstrates feasibility and a signal of efficacy, the format could be scaled into community classes, telehealth modules, or integrated into perinatal services as a nonpharmacologic option for postpartum anxiety.
Watch for recruitment announcements and future pilot results; the next steps will determine whether this short, focused approach can move from protocol to practice. Positive findings would support wider testing and replication, while null results would help refine which mindfulness components or delivery formats work best for parents during the demanding postpartum period.
Know something we missed? Have a correction or additional information?
Submit a Tip

