Study overturns textbook view of CENP-E, reveals chromosome attachment role
CENP-E was recast as an attachment stabilizer, not a chromosome motor, in a finding that upends two decades of mitosis teaching. The shift could reshape cancer research.
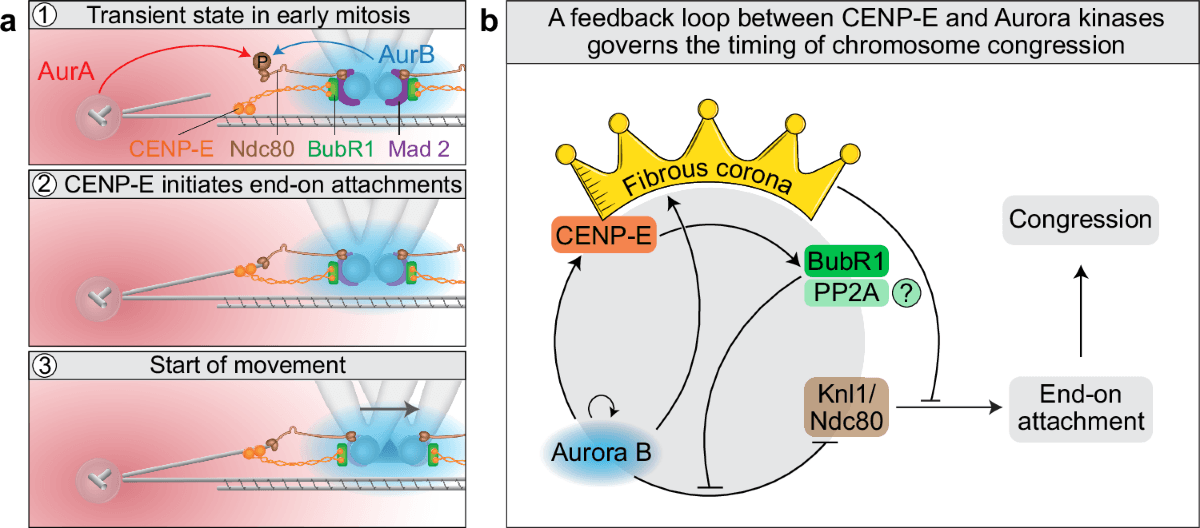
CENP-E was supposed to be the workhorse motor that dragged stray chromosomes to the spindle center. The Ruđer Bošković Institute team in Zagreb says that story was wrong: the protein’s key job is to stabilize the first end-on chromosome-microtubule attachments, a change that rewrites how cell biologists think about segregation errors in cancer and genetic disease.
The study, published in Nature Communications on 21 October 2025, is titled “CENP-E initiates chromosome congression by opposing Aurora kinases to promote end-on attachments.” It was led by Kruno Vukušić and Iva M. Tolić, and it argues that CENP-E, also known as kinesin-7, does not mainly power chromosome movement by gliding kinetochores along microtubules. Instead, it initiates congression by promoting the stabilization of end-on attachments, reducing Aurora B-mediated phosphorylation in a BubR1-dependent manner and helping trigger removal of the fibrous corona. That sequence, the paper says, links congression directly to biorientation.

That is the real textbook overturning here. For years, CENP-E was treated as the engine that hauled chromosomes into place. The RBI says this model is now dismantled by two back-to-back studies, with the new work showing that CENP-E is the factor that helps chromosomes attach correctly in the first place. Nature Communications listed the paper as open access, with 9,873 accesses and 6 citations on the page shown.
The result builds on earlier RBI work that had already started pulling CENP-E out of the simple motor box. A 2023 Nature Communications paper showed that Aurora A and Aurora B phosphorylate CENP-E to release it from autoinhibition, and proposed the Aurora A/B-CENP-E axis as a critical mechanism for fibrous-corona disassembly. A 2021 Communications Biology study went even further from the classic model, showing a non-canonical, kinetochore-independent role for CENP-E in centrosome integrity and orientation of cell division. That work reported PCM1 accumulation, abrogated Plk1 localization and oblique cell divisions when CENP-E was lost.

The broader stakes are not academic. The RBI said chromosome-segregation errors are tied to many cancers and genetic diseases, which is why the new model matters beyond one protein family or one spindle checkpoint debate. The institute described Vukušić as a researcher trained in an ERC Synergy team and preparing to launch his own group, while Tolić heads the Laboratory for Cell Biophysics at RBI and has received two ERC grants. An accompanying image of a metaphase spindle from a human ovarian cancer cell drives home the point: if CENP-E is really a stabilizer of first contact, not a hauling motor, the biology of faulty division may need to be redrawn from the start.
Know something we missed? Have a correction or additional information?
Submit a Tip