Ketamine Combined With Mindfulness Therapy Shows Promise for Opioid Disorder
A new trial found that adding ketamine injections to an 8-week mindfulness program significantly cut drug use and cravings in opioid disorder patients, with zero serious adverse events.
A single-blinded clinical trial registered as NCT04892251 assessed the safety and preliminary efficacy of ketamine-assisted psychotherapy combined with Mindfulness-Oriented Recovery Enhancement for patients with opioid use disorder. The results, published in Nature Mental Health, add a striking data point to a field that has long searched for ways to push past the limits of medication alone.
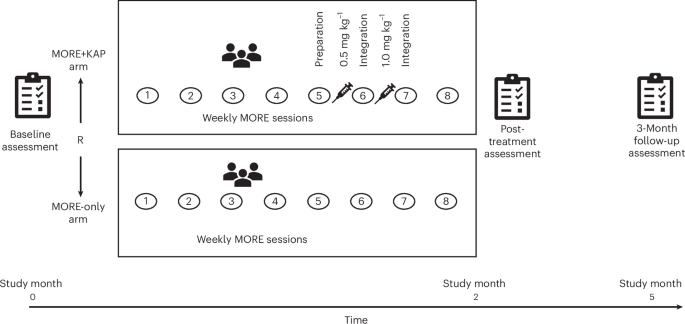
People receiving buprenorphine for OUD were randomized, with 68 participants assigned to either MORE plus ketamine-assisted psychotherapy or MORE only. The MORE arm involved 8 weeks of mindfulness, reappraisal, and savoring training delivered by telehealth videoconferencing. That delivery method matters practically: participants were not required to travel to a clinic for the mindfulness component, which reduces one of the most common barriers to sustained engagement in structured recovery programs.
The ketamine component followed a specific escalating protocol. The MORE plus ketamine arm received a 0.5 mg/kg intramuscular ketamine dose after week 5, then one week later received a dose up to 1.0 mg/kg. The two-injection sequence, timed to the back half of the mindfulness training, was designed to test whether ketamine could augment the psychological work already underway rather than function as a standalone intervention.
The primary outcome was instances of drug use, while secondary outcomes included craving, distress, affective states, and buprenorphine use. The trial demonstrated significant reductions in drug use and cravings without any serious adverse events. No serious ketamine-related adverse events occurred.

The trial was led by Eric L. Garland, whose body of work with MORE spans more than a decade. MORE is an integrative intervention designed to ameliorate addiction, chronic pain, and psychiatric symptoms, combining mindfulness training with cognitive reappraisal and savoring techniques that target the brain's reward processing. MORE has previously demonstrated efficacy in reducing opioid use and craving, hypothetically by restructuring the salience of drug and natural rewards. The ketamine trial represents an attempt to accelerate or deepen that mechanism by pairing the neuroplasticity window opened by ketamine with an active mindfulness curriculum.
Interest in ketamine as a novel treatment for substance use disorders has been increasing due to its NMDA glutamate receptor antagonism and mounting evidence that glutamate neurotransmission is involved in the pathogenesis of both depression and addictions. What makes the Garland trial distinct is the specificity of its pairing: not simply ketamine alongside generic therapy, but ketamine timed within a manualized mindfulness program that has its own established evidence base for opioid populations.
The study's clean safety profile will likely be the detail that opens clinical doors. Prior to this work, it had remained unknown whether combining ketamine treatment with mindfulness-based relapse prevention would lead to different outcomes in the OUD population. That question now has a preliminary answer, and it points toward further investigation at larger scale.
Know something we missed? Have a correction or additional information?
Submit a Tip